The lowest four energy levels for atomic vanadium (V) have the following energies and degeneracies: What is
Question:
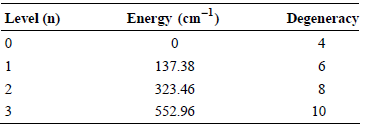
What is the contribution to the average energy from electronic degrees of freedom for V when T = 298 K?
Transcribed Image Text:
Level (n) Energy (cm-1) Degeneracy 4 137.38 6 323.46 552.96 3 10
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
Converting to J U N 143 cm 1 hc nN A 143 cm 1 662610 34 j s 30010 10 cm s 1 n ...View the full answer

Answered By

Sumit kumar
I am an experienced online essay writer with a thorough understanding of any curriculum.and subject expert at Chegg for mathematics, CS subjects..
4.90+
5+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Why do electronic degrees of freedom generally not contribute to the constant volume heat capacity?
-
The three lowest energy levels for atomic carbon (C) have the following energies and degeneracies: Determine the electronic contribution to C V for atomic C at 100. K. Level (n) Energy (cm)...
-
The three lowest energy levels for atomic carbon (C) have the following energies and degeneracies: What is the contribution to the average molar energy from the electronic degrees of freedom for C...
-
A restaurant records the following data over a month for its food: Opening inventory: $31,000 Purchases: $88,000 Closing Inventory: $28,000 Transfers in: $800 Transfers out: $200 Employee meals:...
-
In Exercises 1-2, find the center, foci, and vertices of the hyperbola. Then sketch the hyperbola using the asymptotes as an aid 1. (x - 2)2 / 16 - (y + 1)2 / 9 = 1 2. (x - 1)2 / 144 (7 - 4)2 / 25 = 1
-
In an increasingly competitive world, especially in terms of sales, ethics and ethical conduct by salespeople is of great importance. How would you explain the principles of ethics to newly recruited...
-
Presented here is information for Telco, Inc., for the year ended December 31,2010. Requirements 1. What is the beginning stockholders equity of Telco, Inc.? 2. What is the ending stockholders equity...
-
At the beginning of the tax year, Lizzie holds a $10,000 stock basis as the sole shareholder of Spike, Inc., an S corporation. During the year, Spike reports the following. Determine Lizzies stock...
-
You are considering two independent projects, project A and project B. the required rate of return is 12 percent on each. The expected free cash flow from each project are as follows. Year Project...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Consider an ensemble of units in which the first excited electronic state at energy ε 1 is m 1 -fold degenerate, and the energy of the ground state is m0 -fold degenerate with energy...
-
Consider the following table of diatomic molecules and associated rotational constants: a. Calculate the rotational temperature for each molecule. b. Assuming that these species remain gaseous at 100...
-
Enthalpy can be expressed as (a) \(H=U-P V\) (b) \(H-U=P V\) (c) \(H=A-T S\) (d) \(H-A=T S\).
-
Do you feel that using a project type software is beneficial in your project? Have you used MS project before? What other tools have you used in order to keep you on track of project tasks, lengths...
-
The following information relates to the only product sold by Mastrolia Manufacturing: Sales price per unit Variable cost per unit Fixed costs per year Required: $ 120 72 800,000 a. Compute the...
-
Peter Drucker indicated that it might be a waste of resources for organisations to invest in a training programme that is either unnecessary or inaccurate. Critically examine situations that may...
-
Currently Digby is paying a dividend of $13.44 (per share). If this dividend were raised by $3.64, given its current stock price what would be the Dividend Yield?
-
We're going to create a Google Sheet showing the value of an investment in your future. Well compare two different scenarios: Account A) Investing $1000 when you are 20 years old into an account...
-
When a partners share of partnership liabilities increases, that partners bsis in the partnership interest: a. Increases by the partners share of the liabilities. b. Decreases by the partners share...
-
Accounting policies and practices that are most important to the portrayal of the companys financial condition and results, and require managements most difficult, subjective, or complex judgments...
-
Determine the electron configuration for each of the following atoms: a. Carbon b. Oxygen c. Boron d. Fluorine e. Sodium f. Aluminum
-
In each case, identify the more stable anion. Explain why it is more stable. (a) (b) (c) vs. N. vs. -zo
-
Atropine, extracted from the plant Atropa belladonna, has been used in the treatment of bradycardia (low heart rate) and cardiac arrest. Draw the enantiomer of atropine: CH 0= -
-
1. As part of your co-op placement, you work for a construction company. The boss knows you work fast on computers and asks you to enter all the client information in the database. That evening at...
-
How did railroads change America? Industrialization and Technological Innovation ? Discuss one positive and one negative consequence of American industrialization
-
Why can we say that technological innovation was just as significant in building American cities as it was in driving American industrialization? In what ways did the Civil War help American...

Study smarter with the SolutionInn App


