The molar heat capacity of anhydrous potassium hexacyanoferrate(II) varies with temperature as follows: Calculate the molar enthalpy
Question:
The molar heat capacity of anhydrous potassium hexacyanoferrate(II) varies with temperature as follows:
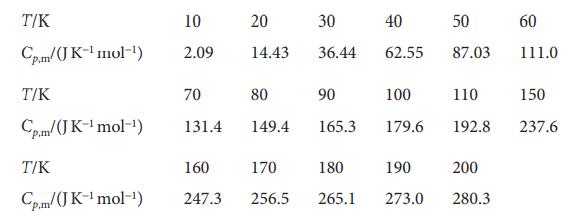
Calculate the molar enthalpy relative to its value at T=0 and the Third-Law entropy at each of these temperatures.
Transcribed Image Text:
T/K Cp.m/(JK-¹ mol-¹) T/K Cp.m/(JK-¹ mol-¹) T/K Cp.m/(JK-¹ mol-¹) 10 2.09 70 131.4 20 14.43 30 36.44 80 149.4 165.3 90 160 170 180 247.3 256.5 265.1 40 62.55 100 179.6 190 273.0 50 87.03 110 192.8 200 280.3 60 111.0 150 237.6
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Answer At T0 the molar enthalpy is 0 and the ThirdLaw entropy ...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
The heat capacity of anhydrous potassium hexacyanoferrate (II) varies with temperature as follows: TIK Cp,m/J K-I mol-I) TIK Cp,m/JK-I mol-I) 10 2.09 100 179.6 20 14.43 110 192.8 30 36.44 150 237.6...
-
The molar heat capacity of lead varies with temperature as follows: TIK 10 15 20 25 30 50 Cp,m/O K-1 rnol-1) 2.8 7.0 10.8 14.1 16.5 21.4 TIK 70 100 150 200 250 298 Cp,m/OK-1 mol-1) 23.3 24.5 25.3...
-
The molar heat capacity of a diatomic molecule is 29.1 J/K mol. Assuming the atmosphere contains only nitrogen gas and there is no heat loss, calculate the total heat intake (in kilojoules) if the...
-
Suggest two reasons why the adjustments proposed by independent auditors more often than not call for reducing recorded earnings.
-
On January 1, 2016, Super View Video, Incorporated issued $ 1,550,000 of $ 1,000 par value, 8%, six- year bonds. Interest is payable semiannually each January 1 and July 1 with the first interest...
-
The thin square plate is suspended from a socket (not shown) which fits the small ball attachment at O. If the plate is made to swing about axis A-A, determine the period for small oscillations....
-
On June 28, 1997, in Las Vegas, heavyweight boxers Mike Tyson and Evander Holyfield met for what proved to be a night to remember. During the third round of the fight, a desperate Tyson illegally bit...
-
To facilitate a move toward JIT production, AB Company is considering a change in its plant layout. The plant controller, Anita Bentley, has been asked to evaluate the costs and benefits of the...
-
Following are the results from two different simple regression analyses, estimating the costs of the purchasing department using number of purchase orders and number of vendors as potential cost...
-
Alpine Thrills Ski Company recently expanded its manufacturing capacity. The firm will now be able to produce up to 15,000 pairs of cross-country skis of either the mountaineering model or the...
-
Account for deviations from Troutons rule for liquids such as water and ethanol. Is their entropy of vaporization larger or smaller than 85 JK 1 mol 1 ? Why?
-
Which of F 2 (g) and I 2 (g) is likely to have the higher standard molar entropy at 298K?
-
Use the method of Examples 7 and 8 to find the Jordan normal form J of each coefficient matrix A given in Problems 23 through 32 (respectively). Example 7 In Example 3 we saw that the matrix ^=[03] A...
-
A ball of mass m = 0.25 kg is moving in the x direction with an initial velocity of vo= 11 m/s. It strikes a ball of m = 2.1 kg initially at rest and sticks together. Find the final velocity for the...
-
Please explain these questions, thanks? Problem Four Jane purchased a residential property on 13 August 1989 (settlement took place on 29 October 1990) for $330,000. Jane borrowed most of the...
-
Albinism in human is inherited as a simple recessive trait. Construct a pedigree for the families below. Indicate the genotypes of the parents and offspring. a) Two normal parents have five children...
-
Two identical 1.60 kg masses are pressed against opposite ends of a spring of force constant 1.80 N/cm, compressing the spring by 15.0 cm from its normal length.
-
On October 1, 20Y8, Jacinto Suarez and Tricia Fritz form a partnership. Suarez agrees to invest $25,000 in cash and inventory valued at $60,000. Fritz invests certain business assets at valuations...
-
A unity feedback system of the form shown in Figure 10.1(a) has a plant (a) Determine the step response when Gc(s) = l, and calculate the settling time and steady state for a ramp input r(t) = t, t >...
-
Evaluate how many lines there are in a true rotational spectrum of CO molecules whose natural vibration frequency is w = 4.09 1014 s1 and moment of inertia I = 1.44 1039 g cm2.
-
At 300 K, the partial vapour pressures of HCl (that is, the partial pressure of the HCl vapour) in liquid GeCl 4 are as follows: Show that the solution obeys Henrys law in this range of mole...
-
The partial molar volumes of acetone (propanone) and chloroform (trichloromethane) in a mixture in which the mole fraction of CHCl 3 is 0.4693 are 74.166 cm 3 mol 1 and 80.235 cm 3 mol 1 ,...
-
Give the ground-state electron configurations of (a) CO, (b) NO, and (c) CN .
-
Jerome, Alwyn, and Floyd have decided to incorporate their partnership. They share profits in the ratio 30:40:30. The noncash assets of the firm are revalued to their fair values. The firm recognizes...
-
Assessing accrual quality of a firm financial statements is important for analysts. Why? calculate the accrual quality of a firm as your own experience.
-
you will be playing a game called Bank on It. The game is located on the AICPA web site. In order to play the game you will be required to register using the instructions below. The game will take...

Study smarter with the SolutionInn App


