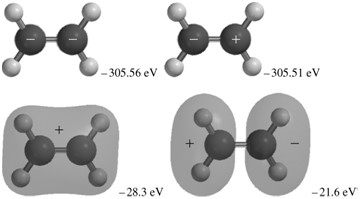
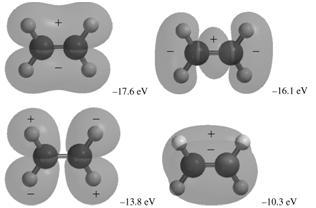
The occupied MOs of ethene are shown next along with the MO energies. Indicate which AOs are
Question:
Transcribed Image Text:
+. -305.51 eV -305.56 eV -28.3 eV -21.6 eV -17.6 eV -16.1 ev -13.8 eV -10.3 eV
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The MOs corresponding to the energies 30356 eV and 30351eV are the inphase and outofphase combinatio...View the full answer

Answered By

Mercy Kangai
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past seven years. I can bring value to your business and help solve your administrative assistant issues. I have extensive experience in marketing and small business management.
4.80+
27+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The occupied MOs of ammonia are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as localized or...
-
The occupied MOs of hydrogen cyanide are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as...
-
Indicate the type of solute-solvent interaction (Section 11.2) that should be most important in each of the following solutions: (a) CCl4 in benzene (C6H6), (b) Methanol (CH3OH) in water, (c) KBr in...
-
Juanita Torres is 35-years old and lately has been working with her financial planner. She is attempting develop a long-term savings and investment program. She has been thinking about all the major...
-
When f (t) = sin t and g (t) = cos t, is h (t) = f (t) g (t) even, odd, or neither? Explain.
-
What is the due diligence, and how can you utilize it when buying a business?
-
How do CIs relate to the specification tree?
-
Identify the working capital accounts related to (a) Revenues recognized and deferred, (b) Cost of goods sold, (c) Employee salary and wages, and (d) Income tax expense. For each account, indicate...
-
Create a complete Java program that will store 20 numbers in an array. The array name is arrNumbers to store the following values: {1,3,5,7,9,8,6,4,2,0,2,4,6,8,9,7,5,3,1} The program will allow user...
-
Casper sells mattresses online. Which sources of Cost Advantage does the company leverage? How does Casper apply the Consumption Chain to help with customer segmentation?
-
Predict whether the ground state or the first excited state of CH 2 should have the larger bond angle on the basis of the Walsh correlation diagram shown in Figure 24.11. Explain your answer. 1b, 17u...
-
Use the geometrical construction shown in Example Problem 24.8 to derive the electron MO levels for cyclobutadiene. What is the total energy of the molecule? How many unpaired electrons will the...
-
An existing corporation that operates a profitable retail business is considering expanding its activities to include manufacturing. The expansion business can be organized in either of two basic...
-
The adjusted trial balance for Chiara Company as of December 31 follows. Cash Debit $ 107,600 Credit Accounts receivable Interest receivable Notes receivable (due in 90 days) Office supplies...
-
Facts John Anderson's property suffered extensive damage in Year 5 as the result of a severe hurricane. The federal government declared the area in which John lived a major disaster area. John has...
-
A manufacturer's Incomplete T-account for Work In Process Inventory follows. Work in Process Inventory Debit Credit Beginning 16,560 Direct materials 24,600 Direct labor 7,320 Overhead applied 2,620...
-
Ryan wants to show the height of a cliff. He is standing at the top of a cliff. He says that he can figure out how high the cliff is by throwing a rock. If he throws the rock with a horizontal...
-
Following are the issuances of stock transactions. 1. A corporation issued 4,000 shares of $5 par value common stock for $35,000 cash. 2. A corporation issued 2,000 shares of no-par common stock to...
-
Access the 2017 Annual Report for Facebook, Inc., and answer the following questions. a. Using information from the companys Income Statement and Income Taxes footnote, what was the companys...
-
In the simple quantity theory of money, what will lead to an increase in aggregate demand? In monetarism, what will lead to an increase in aggregate demand?
-
At 518C, the half-life for the decomposition of a sample of gaseous acetaldehyde (ethanal) initially at 363 Torr was 410 s. When the pressure was 169 Torr, the half-life was 880 s. Determine the...
-
The second-order rate constant for the reaction is 0.11 dm 3 mol 1 s 1 . What is the concentration of ester after (a) 10 s, (b) 10 min when ethyl acetate is added to sodium hydroxide so that the...
-
Sucrose is readily hydrolysed to glucose and fructose in acidic solution. The hydrolysis is often monitored by measuring the angle of rotation of plane polarized light passing through the solution....
-
As you know, the value of. It is defined as the ratio of the circumference of a circle C divided by its diameter 2r. That is x-C/2. a) Let's assume that you measured a circumference of a circle to be...
-
Figure 3 presents a network where N users are sharing a link of 1 Mbps bandwidth. Users are generating data at a rate of 100 kbps when busy, but are busy generating data only with probability p=0.1....
-
The big mall near Sally and Amy's city has extended their operating hours to accommodate the huge crowds expected for the After Thanksgiving Sale. In their eagerness to beat the crowd, Sally forgets...

Study smarter with the SolutionInn App


