The standard potential of the AgCl/Ag,Cl couple has been measured very carefully over a range of
Question:
The standard potential of the AgCl/Ag,Cl− couple has been measured very carefully over a range of temperature (R.G. Bates and V.E. Bowers, J. Res. Nat. Bur. Stand. 53, 283 (1954)) and the results were found to fit the expression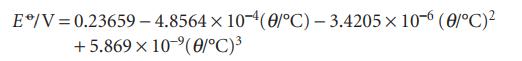
Calculate the standard Gibbs energy and enthalpy of formation of Cl−(aq) and its entropy at 298 K.
Transcribed Image Text:
E/V=0.23659-4.8564 × 10-4 (0/°C) -3.4205 x 10-6 (0/°C)² +5.869 x 10 ⁹(0/°C)³
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Standard potential of the AgClAgCl couple as per the fit expression E o V 023659 48564 x 10 4 x 298 ...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The standard potential of the Zn2+/ Zn electrode is -0.76 V at 25e. The exchange current density for H+ discharge at platinum is 0.79 mA cm-2 Can zinc be plated on to platinum at that temperature?...
-
The standard enthalpy of formation of H2O(l) at 298 K is 285.8 kJ/ mol. Calculate the change in internal energy for the following process at 298 K and 1 atm: H2O(l) H2(g) + O2(g) Eo = ?
-
At 298 K the standard enthalpy of combustion of sucrose is -5797 k] mol-I and the standard Gibbs energy of the reaction is -6333 k] mol ". Estimate the additional non-expansion work that may be...
-
A rectangular loop of wire 24 cm by 72 cm is bent into an L shape, as shown in FIGURE 23-49. The magnetic field in the vicinity of the loop has a magnitude of 0.035 T and points in a direction...
-
Consider the following set of observations: You should not assume these data come from a normal distribution. Test the hypothesis that these data come from a distribution with a median equal to 4....
-
What is operating leverage, and how does it affect a firm's business risk? Show the operating break even point if a company has fixed costs of $200, a sales price of $15, and variables costs of $10.
-
What evidence is protected from disclosure based on a claim of privilege?
-
Refer to Problem 8.3. If the final destination is New Delhi, India, and there is a 30% import tax, which firm should you choose? In Problem 8.3, you have been asked to analyze the bids for 200...
-
A company paid $217.50 for an item. The original price was $296.60, but this was marked down 40%. If the operating expenses are 35% of the cost, find the operating loss and the absolute loss What is...
-
Tanya is a Financial Adviser. One of her clients takes out a life insurance premium at the cost of $276 per year, of which 2.5% is paid as a commission to Tanya. What amount of money would Tanya...
-
The emf of the cell Ag|AgI(s)|AgI(aq)|Ag is +0.9509 V at 25C. Calculate (a) The solubility product of AgI and (b) Its solubility.
-
If the mitochondrial electric potential between matrix and the intermembrane space were 70 mV, as is common for other membranes, how much ATP could be synthesized from the transport of 4 mol H + ,...
-
As the owner of a business, you must make an investment decision. The investment will expand your company's production plant at a cost of $1 million. The expansion will generate income of $150,000...
-
Partial balance sheets and additional information are listed below for Sowell Company.Sowell Company Partial Balance Sheets as of December 31 2024 2023 Assets Cash$ 35,000$ 15,500 Accounts receivable...
-
Concord Company has the following account balances: Sales Revenue $ 2 1 1 , 4 0 0 , Sales Discounts $ 4 , 3 1 0 , Cost of Goods Sold $ 1 3 8 , 6 0 0 , and Inventory $ 4 3 , 9 0 0 . Prepare the...
-
Suppose the graph of f is given. Describe how the graph of each function can be obtained from the graph of f. List the transformations in the correct order! a. 2f(x+5)-1 Transformation 1:...
-
Arctic Wines sold $680,000 in wines during their fall color season in October. Given the following information, calculate the Arctic Wine's gross margin and gross margin percentage. Use the...
-
Write a program that converts a temperature from Fahrenheit to Celsius. It should do the following: Prompt the user for input Read adouble value from the keyboard Calculate the result.Here is the...
-
Prepare a process flow chart for the cheque processing operation. How many of the steps are value-adding?
-
How has the globalization of firms affected the diversity of their employees? Why has increased diversity put an additional burden on accounting systems?
-
In a double-glazed window, the panes of glass are separated by 1.0 cm. What is the rate of transfer of heat by conduction from the warm room (28 C) to the cold exterior (15 C) through a window of...
-
A lump of sucrose of mass 10.0 g is suspended in the middle of a spherical flask of water of radius 10 cm at 25 C. What is the concentration of sucrose at the wall of the flask after (a) 1.0h, (b)...
-
Confirm that is a solution of the diffusion equation with convection (eqn 19C.10) with all the solute concentrated at x=x 0 at t=0 and plot the concentration profile at a series of times to show how...
-
A potato is launched out of a building (building A) from the 10th floor which is 15 m high, with a velocity of 55 m/s at an angle of 35.0 above the horizontal. There is a building (building 2) 130 m...
-
An eagle is flying horizontally at a speed of 2.9 m/s when the fish in her talons wiggles loose and falls into the lake 4.4 m below. Calculate the magnitude of the velocity of the fish relative to...
-
Because of its high dielectric strength, SF6 (sulfur hexafluo-ride) gas is widely used as an insulator and a dielectric in HV applications such as HV transform-ers, switches, circuit breakers,...

Study smarter with the SolutionInn App


