Use the phase diagram in Fig. 6.40 to state (a) The solubility of Ag in Sn at
Question:
Use the phase diagram in Fig. 6.40 to state
(a) The solubility of Ag in Sn at 800°C and
(b) The solubility of Ag3Sn in Ag at 460°C,
(c) The solubility of Ag3Sn in Ag at 300°C.
Fig. 6.40
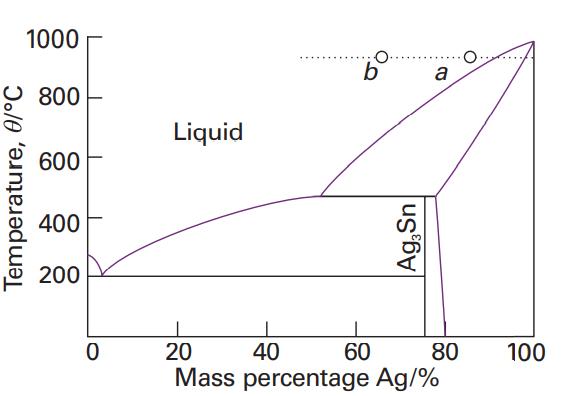
Transcribed Image Text:
Temperature, 0/°C 1000 800 600 400 200 0 Liquid O. b Ag,Sn 60 a 20 40 80 Mass percentage Ag/% 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
Solution a The solubility of Ag in Sn at 800C is given by x...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the phase diagram in Fig. 6.41 to state (a) The solubility of B in A at 500C and (b) The solubility of AB2 in A at 390C, (c) The solubility of ABz in Bat 300e.
-
The phase diagram for neon is Temperature (K) Use the phase diagram to answer the following questions. (a) What is the approximate value of the normal melting point? (b) Over what pressure range will...
-
Use the phase diagram in Fig. 5.3 to state (i) the solubility of Ag in Sn at 800 C and (ii) the solubility of Ag 3 Sn in Ag at 460 C, (iii) the solubility of Ag 3 Sn in Ag at 300 C. Data in Fig. 5.3...
-
Passenger table (passengerid, address etc.) Flight table (flight id, departure, destination, depDate) Booking table (cID, fid, date, cost) a. Find Passengers who live in Chicago b. Total number of...
-
What is marking-to-market for a future? Why is this marking-to-market important for reducing counterparty risk?
-
If three different orders are selected, find the probability that they are all not accurate. Use the data in the following table, which lists drive-thru order accuracy at popular fast food chains...
-
What do you know about the mathematical value of a project's internal rate of return under each of the following conditions? a. The present worth of the project is greater than 0 . b. The present...
-
Kuchin Company reports the following for the month of June. Instructions(a) Compute the cost of the ending inventory and the cost of goods sold under (1) FIFO,(2) LIFO, and (3) average-cost.(b) Which...
-
A square insulating plate, 2 meters by 2 meters, lies flat on the floor. A total charge Q = 8 10-8 C is distributed uniformly on the sheet. A pith ball (a small non-conducting ball made of a light...
-
Withers Company uses a standard process costing system in its one production department. Material A is added at the beginning of the process, and Material B is added when the units are 90% complete....
-
Show that two phases are in mechanical equilibrium only if their pressures are equal.
-
Indicate on the phase diagram in Fig. 6.41 the feature that denotes incongruent melting. What is the composition of the eutectic mixture and at what temperature does it melt? Fig. 6.41 Temperature,...
-
1. Explain why assigning a devils advocate during a decision-making meeting reduces groupthink. 2. Give an example of a decision you have been involved in that might have benefited from the...
-
Fast Jack is a fast-food restaurant that employs minors. Fast Jack is subject to the federal child labor, minimum wage, and overtime provisions in a. the Family and Medical Leave Act. b. the...
-
What are some protections afforded by the federal age-discrimination law?
-
National Supplies Company hires Linda and Brad as employees to deal with third-party purchasers and suppliers. Linda and Brad are a. principals. b. agents. c. both a and b. d. none of the above.
-
Discrimination complaints under federal law must be filed with the Equal Opportunity Employment Commission. (True/False)
-
When can an employer legally hire someone not authorized to work in this country?
-
Explain and demonstrate how replacement cost accounting affects reported profit as compared with historical cost accounting.
-
Using the parallel-axis theorem, determine the product of inertia of the area shown with respect to the centroidal x and y axes. 6 in. 9 in. 9 in- 4.5 in. in. 4.5 in.
-
How is the method of combination differences used in rotation vibration spectroscopy to determine rotational constants?
-
The wavenumber of the J=32 rotational transition of 1 H 35 Cl considered as a rigid rotor is 63.56 cm 1 ; what is the HCl bond length?
-
Classify the following rotors: (i) O 3 , (ii) CH 3 CH 3 , (iii) XeO 4 , (iv) FeCp 2 (Cp denotes the cyclopentadienyl group, C 5 H 5 ).
-
1.Contracts for the sale of the two properties were initially exchanged during the year ended 30 June 2005, and eventually were finalised before the end of 2005, so why were the auditors criticised...
-
You just won the "NoLuck" lottery, and you selected the one lump sum option. You plan to use it for 25 years and make quarterly withdrawals of $50,000. The APR is S percent a year, compounded...
-
Albee, Inc. issued $100,000 face value bonds payable on September 1, 2018 at a price of $92,278. The bonds carried a stated annual interest rate of 8%, payable semi-annually on September 1 and March...

Study smarter with the SolutionInn App


