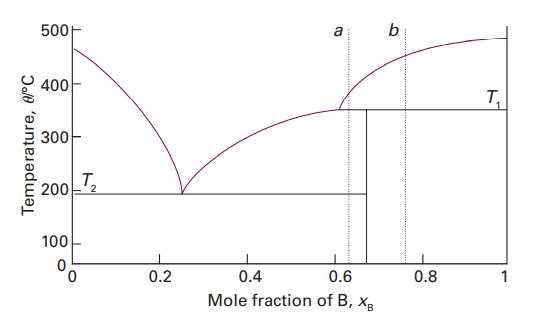
Use the phase diagram in Fig. 5.3 to state (i) the solubility of Ag in Sn at
Question:
Use the phase diagram in Fig. 5.3 to state (i) the solubility of Ag in Sn at 800 °C and (ii) the solubility of Ag3Sn in Ag at 460 °C, (iii) the solubility of Ag3Sn in Ag at 300 °C.
Data in Fig. 5.3
Transcribed Image Text:
Temperature, 0/°C 500 400 300 200 100 0 I _T₂ 0.2 a 0.4 0.6 Mole fraction of B, XB b 0.8 T₁ 1 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
Answer i The solubility of Ag i...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Use the phase diagram in Fig. 6.41 to state (a) The solubility of B in A at 500C and (b) The solubility of AB2 in A at 390C, (c) The solubility of ABz in Bat 300e.
-
The phase diagram for neon is Temperature (K) Use the phase diagram to answer the following questions. (a) What is the approximate value of the normal melting point? (b) Over what pressure range will...
-
Use the phase diagram in Fig. 6.40 to state (a) The solubility of Ag in Sn at 800C and (b) The solubility of Ag 3 Sn in Ag at 460C, (c) The solubility of Ag 3 Sn in Ag at 300C. Fig. 6.40 Temperature,...
-
Rainbow manufactures wooden backyard playground equipment. Rainbow estimated $1,785,000 of manufacturing overhead and $2,100,000 of direct labour cost for the year. After the year was over, the...
-
Fredrick Wilson Company, an IFRS reporter, determined that one of its finite- life intangible assets is impaired. The assets net carrying value on the date of the impairment is $ 905,000. Fredrick...
-
Charles Wong, the owner of Original Woods Inc. (OWI), would like to become a strategic partner in SIL. SIL currently purchases 75% of its wood materials from OWI, so this partnership would streamline...
-
How much air is needed to fully combust fuel oil \(\left(\mathrm{CH}_{1.67} \mathrm{O}_{0.006} \mathrm{~N}_{0.005} \mathrm{~S}_{0.002} ight)\) if \(25 \%\) excess air is required?
-
Sadowski Video Center accumulates the following cost and market data at December 31. Compute the lower-of-cost-or-market valuation for Sadowskiinventory. Inventory Categories Cameras Camcorders DVDs...
-
Jane Dutton, a co-founder of "Positive Organisational Scholarship" (POS), spoke to us. How did she describe POS in her talk? Refer to the video between 15:30 to 25:06 minutes:...
-
Gerrard Construction Co. is an excavation contractor. The following summarized data (in thousands) are taken from the December 31, 2019, financial statements: At December 31, 2018, total assets were...
-
The temperaturecomposition diagram for the Ca/Si binary system is shown in Fig. 5.7. (a) Identify eutectics, congruent melting compounds, and incongruent melting compounds. (b) If a 20 per cent by...
-
Referring to Fig. 5.8, deduce the molar solubility of (i) NH 4 Cl, (ii) (NH 4 ) 2 SO 4 in water at 25 C. Data in Fig. 5.8, 0.8 NHCI 10 0.6 0.2 0.44 0.2 P=2 0/1 HO P=1 fg P=3 0.4 0.8 P=2 0.6 0.6 0.4...
-
1. Identify the key elements in McDonalds global marketing strategy. Despite a slowdown in global fast-food consumption, McDonalds continues to be a success story. What is the key? Does McDonalds...
-
What are the primary challenges of online meetings, according to research?
-
Why should a team develop a charter?
-
Ogden and Richardss semantic triangle contains three elements: the symbol, the reference, and the referent. What does each element represent?
-
What are some common elements of a high performing teams culture?
-
When is it especially beneficial to consider holding a meeting online, instead of face-to-face?
-
Predict the signs of H, S, and G of the system for the following processes at 1 atm: (a) ammonia melts at 260C, (b) ammonia melts at -77.7C, (c) ammonia melts at -100C. (The normal melting point of...
-
Create a data model for one of the processes in the end-of-chapter Exercises for Chapter 4. Explain how you would balance the data model and process model.
-
Discuss the following statement: If the temperature of the system increased, heat must have been added to it.
-
Oxygen reacts with solid glycylglycine C 4 H 8 N 2 O 3 to form urea CH 4 N 2 O, carbon dioxide, and water: 3O 2 (g) + C 4 H 8 N 2 O 3 (s) CH 4 N 2 O(s) + 3CO 2 (g) + 2H 2 O(l) At T = 298 K and 1.00...
-
Discuss the following statement: Heating an object causes its temperature to increase.
-
A proton (mass of 1.67 x 10-27 kg) is moving at 1.25 x 106 m/s directly toward a stationary helium nucleus (mass 6.64 x 10-27 kg). After a head-on elastic collision, what is the velocity of the...
-
The December 31, Year 4, balance sheet for Vernon Corporation is presented here. These are the only accounts on Vernon's balan sheet. Amounts indicated by question marks (?) can be calculated using...
-
Reyes Manufacturing Company uses a job order cost system. At the beginning of January, the company had one job in process (Job 201) and one job completed but not yet sold (Job 200). Job 202 was...

Study smarter with the SolutionInn App


