What do you expect the electronic spectrum to look like for the ground and excited states shown
Question:
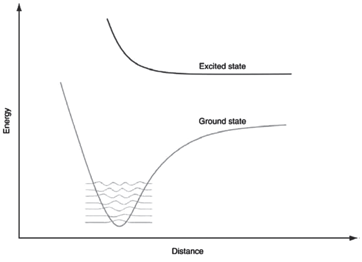
Transcribed Image Text:
Excited state Ground state Distance ABsoug
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
The electronic spectrum will have no ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Do you expect the viscosity of glycerol, C3H5(OH)3, to be larger or smaller than that of 1-propanol, C3H7OH? Explain? (a) Glycerol (b) 1-Propanol
-
Consider the tetrahedral anions VO43- (orthovanadate ion), GrO42- (chromate ion), and MnO4 (permanganate ion). (a) These anions are isoelectronic. What does this statement mean? (b)Would you expect...
-
Do you expect the light given off by (a) a neon sign or (b) an incandescent lightbulb to be continuous in distribution or in the form of a line spectrum? Explain.
-
A manufacturing company reports the following information for the month of May. Note: Assume all raw materials were used as direct materials. Activities for May Advertising expense Raw materials...
-
In Exercises 1-4, find the exact values of the six trigonometric functions of the angle . 1. 2. 3. 4. 8. 13
-
In addition to advice about which business structure David should use, he has asked for advice on how to go about setting up the business. David would like to understand any legal requirements for...
-
Describe various defenses and remedies available for nonperformance of a contract.
-
Ringo loves playing rock n roll music at high volume. Luciano loves opera and hates rock n roll. Unfortunately, they are next-door neighbors in an apartment building with paper-thin walls. a. What is...
-
Image transcription text Requirements Statement for Meeting Scheduling Software: Many ventures both academic and business require constant collaboration to be successful. The most basic form of...
-
A gas at upstream condition (T1 ,P1) is throttled in a downstream pressure of 1.2 bar. Use the Redlich / Kwong equation to estimate the downstream temperature and S of the gas for one of the...
-
Because internal conversion is in general very fast, the absorption and fluorescence spectra are shifted in frequency as shown in Figure 25.10. This shift is crucial in making fluorescence...
-
Why are the spectra of the individual molecules in the bottom trace of Figure 25.15 shifted in frequency? 10,000 Molecules x1 1,000 x10 x100 100 10 Frequency Absorption
-
Write the hexadecimal representation of the following decimal numbers: a. 12 10 b. 160 10
-
Discuss the importance of organisational and national cultures in managing SHRM interventions;
-
As a senior HR manager in a major MNC, what arguments would you anticipate using to defend your company against the anti-globalisation lobbys position that globalisation was disadvantageous to your...
-
Explore the three main perspectives through which culture has been explored within organisations: integration, differentiation and fragmentation;
-
Terry Thompson, who meets all the requirements of Section 1237, sells five lots during 2017 at a gain of \(\$ 30,000\). During 2018 , he sells the sixth lot (basis \(\$ 5,000\) ) at a selling price...
-
The reasons that companies pursue a strategy of internationalisation are not new. Historically, empires have been built on thriving international trade. Yet the rate of growth of international trade...
-
In problem, solve each logarithmic equation. Express irrational solutions in exact form and as a decimal rounded to three decimal places. log 2 (x + 7) + log 2 (x + 8) = 1
-
What is a manufacturing system?
-
For how long on average would an H atom remain on a surface at 298 K if its desorption activation energy were (a) 15 kJ mol 1 , (b) 150 kJ mol 1 ? Take 0 = 0.10 ps. For how long on average would the...
-
The adsorption of a gas is described by the Langmuir isotherm with K = 0.85 kPa 1 at 25C. Calculate the pressure at which the fractional surface coverage is (a) 0.15, (b) 0.95.
-
(a) Discuss the main structural features of the electrical double layer. (b) Distinguish between the electrical double layer and the Nernst diffusion layer.
-
Prove that Russian multiplication does what it needs to do, i.e. the result is the product of the two integers. Do not use the proof of the book. It is mainly an exercise in understanding the binary...
-
What is the fundament difference between growing a unicorn versus a camel? What are the financial recommendations for start-ups and the reasons given for those choices? Do you agree with these...
-
16. Nickel Inc. bought $500,000 of 3-year, 9% bonds as an investment on December 31, 2015 for $545,000. Nickel uses straight-line amortization. On May 1, 2016, $100,000 of the bonds were redeemed at...

Study smarter with the SolutionInn App


