Because internal conversion is in general very fast, the absorption and fluorescence spectra are shifted in frequency
Question:
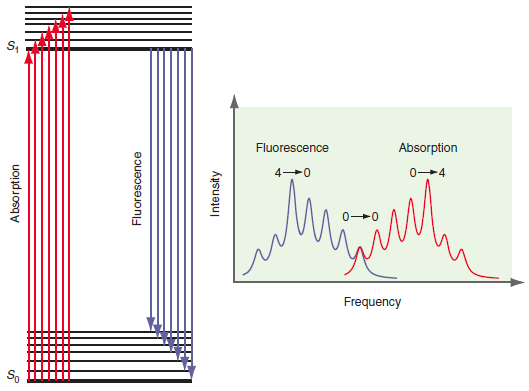
Transcribed Image Text:
Fluorescence Absorption 0-4 Frequency So Absorption Fluorescence Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
In the experiment the sample is constantly being illuminated with light to effect the transition of ...View the full answer

Answered By

Deborah Joseph
My experience has a tutor has helped me with learning and relearning. You learn everyday actually and there are changes that are made to the curriculum every time so being a tutor has helped in keeping me updated about the present curriculum and all.
I have also been able to help over 100 students achieve better grades particularly in the categories of Math and Biology both in their internal examinations and external examinations.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Briefly describe or define (a) Fluorescence. (b) Internal conversion. (c) Stokes shift. (d) Inner-filter effect.
-
The fluorescence spectrum of anthracene vapour shows a series of peaks of increasing intensity with individual maxima at 440 nm, 410 nm, 390 nm, and 370 nm followed by a sharp cut-off at shorter...
-
One of the principal methods for obtaining the electronic spectra of unstable radicals is to study the spectra of comets, which are almost entirely due to radicals. Many radical spectra have been...
-
Dakota Company had net sales ( at retail ) of $ 1 4 4 , 0 0 0 . \ table [ [ , At Cost,At Retail ] , [ Beginning inventory,$ 3 3 , 3 0 0 , $ 6 5 , 6 0 0 es Dakota Company had net sales (at retail) of...
-
In Exercises 1-4, find each value of in degrees (0o < < 90o) and radians (0 < < / 2) without using a calculator. 1. a. sin = 1 / 2 b. csc = 2 2. a. cos = 2 / 2 b. tan = 1 3. a. sec = 2 b....
-
Tim Colley and John Williams had not been in business long as Heritage Research Associates, a small marketing research firm they started in their hometown. They were barely making ends meet by taking...
-
What problems with existing Engineering paradigms is it intended to correct?
-
Brandt Boot Co. sells men's, women's, and children's boots. For each type of boot sold, it operates a separate department that has its own manager. The manager of the men's department has a sales...
-
The exchange rate between euros and dollars is currently $1.03per euro. Annual inflation is expected to be 1.2% in Europe and2.8% in the U.S. Part 1 If relative purchasing power parity holds, what is...
-
Five clowns each have a red wig and a blue wig, which they are all equally likely to wear at any particular time. Find the probability that, at any particular time: a. Exactly two clowns are wearing...
-
Explain why the spectator species M in Equation (25.5) is needed to make the reaction proceed.
-
What do you expect the electronic spectrum to look like for the ground and excited states shown in the figure below? Excited state Ground state Distance ABsoug
-
A firm will pay a $4.00 dividend at the end of year one, has a share price of $50, and a constant growth rate of 5 percent. Compute the required (expected) rate of return.
-
Dallas Company uses a job order costing system. The company's executives estimated that direct labor would be $ 3 , 8 4 0 , 0 0 0 ( 1 6 0 , 0 0 0 hours at $ 2 4 / hour ) and that factory overhead...
-
How have you seen issues of autonomous and positive face create conflict in your experience? What have you done to resolve issues of autonomous and positive face? Are the strategies you use for each...
-
Department R had 4 , 7 0 0 units in work in process that were 7 9 % completed as to labor and overhead at the beginning of the period; 3 7 , 6 0 0 units of direct materials were added during the...
-
Compose and post a thread of at least 500 words , in which you compare and contrast Tita and her two sisters, Rosaura and Gertrudis. Comment specifically on the differences in their personalities,...
-
The following account balances were taken from the 2 0 2 4 post - closing trial balance of the Bowler Corporation: cash, $ 5 , 0 0 0 ; accounts receivable, $ 1 5 , 0 0 0 ; inventory, $ 2 6 , 0 0 0 ;...
-
ATW corporation currently uses the FIFO method of accounting for its inventory for book and tax purposes. Its beginning inventory for the current year was $8,000,000. Its ending inventory for the...
-
7 A 29-year-old, previously healthy man suddenly collapses at a party where legal and illicit drugs are being used. Enroute to the hospital, he requires resuscitation with defibrillation to establish...
-
In an experiment on the Pt|H 2 |H + electrode in dilute H 2 SO 4 the following current densities were observed at 25C. Evaluate and j 0 for the electrode. How would the current density at this...
-
Nitrogen gas adsorbed on charcoal to the extent of 0.921 cm 3 g 1 at 490 kPa and 190 K, but at 250 K the same amount of adsorption was achieved only when the pressure was increased to 3.2 MPa. What...
-
Hydrogen iodide is very strongly adsorbed on gold but only slightly adsorbed on platinum. Assume the adsorption follows the Langmuir isotherm and predict the order of the HI decomposition reaction on...
-
s Machine-hours required to support estimated production Fixed manufacturing overhead cost Variable manufacturing overhead cost per machine-hour Required: 1. Compute the plantwide predetermined...
-
How do visionary leaders navigate the complexities of organizational dynamics to cultivate a culture of innovation and adaptability ? Explain
-
Finnish Corporation manufactures custom molds for use in the extrusion industry. The company allocates manufacturing overhead based on machine hours. Selected data for costs incurred for Job 532 are...

Study smarter with the SolutionInn App


