What is the functional dependence of the 1s orbital energy on Z in Figure 21.7? Check your
Question:
Figure 21.7
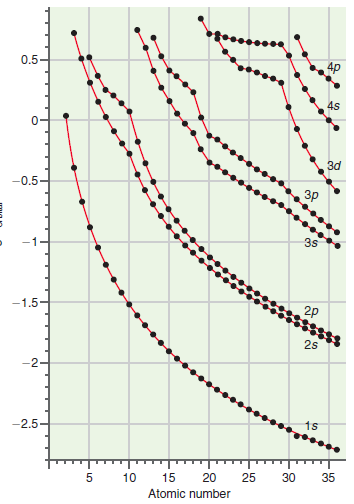
Transcribed Image Text:
0.5 4s Зd -0.5 зр 3s 2p -1.5 2s 15 -2.5 35 30 25 20 10 Atomic number -15
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
The radial equation for a one electron atom with nuclear charge Z ...View the full answer

Answered By

Sidharth Jain
My name is Sidharth. I completed engineering from National Institute of Technology Durgapur which is one of the top college in India. I am currently working as an Maths Faculty in one of the biggest IITJEE institute in India. Due to my passion in teaching and Maths, I came to this field. I've been teaching for almost 3 years.
Apart from it I also worked as an Expert Answerer on Chegg.com. I have many clients from USA to whom I teach online and help them in their assignments. I worked on many online classes on mymathlab and webassign. I guarantee for grade 'A'.
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Electron and hole concentrations increase with temperature. For pure silicon, suitable expressions are h = e = 6200T 1.5 e 7000 /T C/m 3 . The functional dependence of the mobilities on temperature...
-
Extend the considerations of the preceding problem to particle diffusion, and assume that there is a net particle generation rate gu that is proportional to the local particle concentration, gu =...
-
Vortex flow (a) Show that the complex potential w = (iT/2) in z describes the flow in a vortex. Verify that the tangential velocity is given by v = T/2 and that v r = 0. This type of flow is...
-
Brennan Physiotherapies had a beta of 0.85. Reasonable estimates for the RF and the required rate of return on the market, R(Rm) were 7% and 15%, respectively. What is the required rate of return on...
-
A privately owned yacht leaves a dock in Myrtle Beach, South Carolina, and heads toward Freeport in the Bahamas at a bearing of S 1.4 E. The yacht averages a speed of 20 knots over the...
-
This chapter describes HRs changing role in business. Describe each component that is involved in human resource management.
-
Two A-36 steel wires are used to support the 650-lb engine. Originally, AB is 32 in. long and A B is 32.008 in. long. Determine the force supported by each wire when the engine is suspended from...
-
Name five key factors that affect a firms external financing requirements.
-
The current tourism industry booms in Australia had given the HR department at ANIBT Hotel the opportunity to employ a very diverse range of ethnic employees for all levels of the industry. This...
-
Using the Gibbs function data, determine the equilibrium constant KP for the reaction H2 + 1/2 O2 H2O at (a) 298 K and (b) 2000 K. Compare your results with the KP values listed in Table A28.
-
The total energy eigenvalues for the hydrogen atom are given by E n = e 2 / (8Ïε 0 a 0 n), n = 1, 2, 3, 4,¦, and the three quantum numbers associated with the total energy...
-
Why is the total energy of a many-electron atom not equal to the sum of the orbital energies for each electron?
-
Find the most general antiderivative of the function. (Check your answer by differentiation.) f(x) = 10/x 9
-
Revisions to Chinas budget law, passed on 31 August 2014, represent a significant reform, providing a framework for significantly greater transparency and accountability for local government says...
-
The Africa Research Bulletin reported the results of a prefeasibility study by Ironveld plc stating that the grades for pig iron and ferro vanadium were better than expected at its project on the...
-
What is the capacitance of a parallel-plate capacitor that has a plate area \(A\) and a plate separation distance \(d\) ?
-
Increasingly, householders and small businesses are considering renewable power generation systems to decrease their costs. Most of the UK, Ireland and Scandinavia are suited to generating energy...
-
The radius of the dome on one very large Van de Graaff generator is about \(2.5 \mathrm{~m}\), and air breaks down when the field magnitude is about \(3.0 \times 10^{6} \mathrm{~V} / \mathrm{m}\)....
-
Fill in the blank with an appropriate word, phrase, or symbol(s). If a candidate receives a majority of first-place votes in an election, that candidate should be declared the winner. This criterion...
-
6 (a) Briefly develop a mathematical model of the behaviour of a copper-twisted pair cable (b) Derive the magnetic energy from: w given that: K + w, where the - - k symbols have their usual meaning...
-
Calculate the percentage change in K x for the reaction H 2 CO(g) CO(g) + H 2 (g) when the total pressure is increased from 1.0 bar to 2.0 bar at constant temperature.
-
A sealed container was filled with 0.300 mol H 2 (g), 0.400 mol I 2 (g), and 0.200 mol HI(g) at 870 K and total pressure 1.00 bar. Calculate the amounts of the components in the mixture at...
-
The equilibrium constant for the gas-phase isomerization of borneol (C 10 H 17 OH) to isoborneol at 503 K is 0.106. A mixture consisting of 7.50 g of borneol and 14.0 g of isoborneol in a container...
-
A 9 Loan amount Annual interest rate Number of years until maturity Annual payment amount B $300,000.00 Date Graded Worksheet 7% 4 On January 1, 2024, a company borrows cash from a local bank by...
-
As we have seen in this course, virtually every generation has felt that the existing threat to its privacy was unprecedented in nature. That said, the sheer capacity to collect metadata which exists...
-
In 1982 the inflation rate hit 16%. Suppose that the average cost of a textbook in 1982 was $20. What was the expected cost in the year 2017 if we project this rate of inflation on the cost? (Assume...

Study smarter with the SolutionInn App


