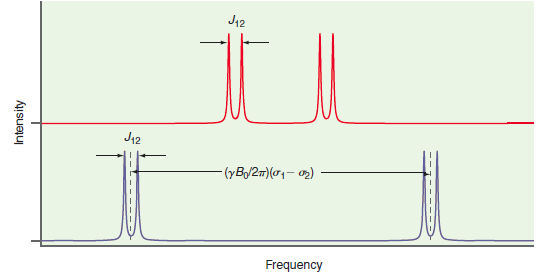
Why are the multiplet splittings in Figure 28.9 not dependent on the static magnetic field? J12 J42
Question:
Transcribed Image Text:
J12 J42 (yB/2m)(01- 2) Frequency Intensity -----
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
The multiplet splitting is determ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Why is the multiplet splitting for coupled spins independent of the static magnetic field?
-
Heat capacity from internal degrees of freedom (a) Consider a two-level system with an energy splitting kB? between upper and lower states; the splitting may arise from a magnetic field or in other...
-
In FIGURE EX10.28, what is the maximum speed a 200 g particle could have at x = 2.0 m and never reach x = 6.0 m? U (J) 16- 12 - 8- 4. 0+ x (m) FIGURE EX10.28 4 2.
-
Products are 1. Lip gloss or lipstick with SPF 2. Custom facial sheets you can make online or in store pickup and have it send to your home based on your needs 3. A foundation sunscreen Price Provide...
-
After discontinuing all advertising for a tool kit in 2010, the manufacturer noted that sales began to drop according to the model S = 500,000 / 1 + 0.1ekt where S represents the number of units sold...
-
What is the value of autonomous consumption? Use the information in the following table Y C G X $100 $120 $20 $30 $10 $300 $300 $20 $30 -$10 $500 $480 $20 $30 -$30 $700 $660 $20 $30 -$50
-
What is a System Storage Concept? Identify examples of its UCs.
-
XYZ is a calendar-year corporation that began business on January 1, 2018. For the year, it reported the following information in its current-year audited income statement. Notes with important tax...
-
What is the difference between diversifiable and non-diversifiable risks? Give examples of each and explain why you placed the risk in that category. If it is diversifiable, what can be done to...
-
Air enters a normal shock at 22.6 kPa, 217 K, and 680 m/s. Calculate the stagnation pressure and Mach number upstream of the shock, as well as pressure, temperature, velocity, Mach number, and...
-
Why does the H atom on the OH group not lead to a multiplet splitting of the methyl hydrogens of ethanol?
-
Redraw Figure 28.2 for β spins. What is the direction of precession for the spins and for the macroscopic magnetic moment? Bo VA VVA
-
Which of the following would be your justification for treating as an asset the amount due from a customer in respect of transport work already carried out? (i) The customer is within our control...
-
Consider a square wire loop that carries a current I = 25 mA in the clockwise direction. The loop has side length a = 1.5 cm. What is the magnetic field B at point p in the center of the square loop?...
-
A student stands at the edge of a cliff and throws a stone horizontally over the edge with a speed of vo = 16.5 m/s. The cliff is h = 48.0 m above a flat, horizontal beach as shown in the figure....
-
On January 1, 2021, Frontier World issues $40.8 million of 7% bonds, due in 10 years, with interest payable semiannually on June 30 and December 31 each year. The proceeds will be used to build a new...
-
The following income statement items appeared on the adjusted trial balance of Schembri Manufacturing Corporation for the year ended December 31, 2024 ($ in thousands): sales revenue, $18,300; cost...
-
The following information is from Ellerby Company's comparative balance sheets. At December 31 Furniture Accumulated depreciation-Furniture Current Year $ 157,500 (97,200) Prior Year $ 218,500...
-
This year Jack intends to file a married-joint return. Jack received $167,500 of salary, and paid $5,000 of interest on loans used to pay qualified tuition costs for his dependent daughter, Deb. This...
-
Why are stocks usually more risky than bonds?
-
The complex ion [Fe(OH 2 ) 6 ] 3+ has an electronic absorption spectrum with a maximum at 700nm. Estimate a value of O for the complex.
-
The R-branch of the 1 u 1 g + transition of H 2 shows a band head at the very low value of J=1. The rotational constant of the ground state is 60.80 cm 1 . What is the rotational constant of the...
-
The highest kinetic energy electrons in the photoelectron spectrum of H 2 O using 21.22 eV radiation are at about 9 eV and show a large vibrational spacing of 0.41 eV. The symmetric stretching mode...
-
Pronghorn Corp reported net sales $675,000, cost of goods sold $438,750, operating expenses $154,000, and net income $69,525. Calculate the profit margin and gross profit rate. (Round Profit margin...
-
Victory Company uses weighted average process costing. The company has two production processes. Conversion cost is added evenly throughout each process. Direct ma added at the beginning of the first...
-
Here are selected 2022 transactions of Oriole Company. Jan. 1 June 30 Dec. 31 Retired a piece of machinery that was purchased on January 1, 2012. The machine cost $64,000 and had a useful life of 10...

Study smarter with the SolutionInn App


