Is it possible for a heat engine to operate as shown in the following diagram? Explain, using
Question:
Is it possible for a heat engine to operate as shown in the following diagram? Explain, using the laws of thermodynamics.
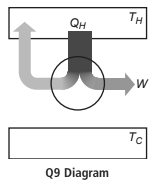
Transcribed Image Text:
QH Q9 Diagram TH W Tc
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
No it is not possible for a heat engine to operate as shown in the figure Heat is t...View the full answer

Answered By

Subhendu Biswas
Hi, i am Subhendu Biswas, assistant teacher of mathematics at a High Secondary Government School. I have done M.Sc , B.Ed in mathematics. I have 8 years experience in mathematics teaching at secondary and high secondary level. I can teach mathematics from calss 9 - 12 ( all boards). I want to share my knowledge and experience in the field of mathematics. Thank You.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physics of Everyday Phenomena A conceptual Introduction to physics
ISBN: 978-0073512112
6th edition
Authors: W. Thomas Griffith, Juliet W. Brosing
Question Posted:
Students also viewed these Physics questions
-
Is it possible for a heat pump to operate as shown in the diagram? Explain, using the laws of thermodynamics. TH Tc QH Qc Q20 Diagram
-
Is it possible for a fluorescent material to emit radiation in the ultraviolet region after absorbing visible light? Explain your answer.
-
Is it possible for a heat engine to operate without rejecting any waste heat to a low-temperature reservoir? Explain.
-
Sketch the graph of the function and state its domain. f (x) = ln |x|
-
Gary, who is single, sells his principal residence (owned and occupied by him for seven years) in November 2013 for a realized gain of $148,000. He had purchased a more expensive new residence eight...
-
Lactones can be prepared from diethyl malonate and epoxides. Diethyl malonate is treated with a base, followed by an epoxide, followed by heating in aqueous acid: Using this process, identify what...
-
Reduced temperature of a pure substance is the ratio of its (a) Critical temperature to temperature (b) Critical temperature to saturated temperature (c) Saturated temperature to critical temperature...
-
Flexible-budget and sales volume variances, market-share and market-size variances. Marron, Inc., produces the basic fillings used in many popular frozen desserts and treats'vanilla and chocolate ice...
-
Novak Corp. owns and operates a number of outdoor clothing retail stores in the Pacific Northwest. Recently, the company has decided to locate another store in a rapidly growing area of Idaho. The...
-
How do you feel about having management responsibilities in today's world, characterized by uncertainty, ambiguity, and sudden changes or threats from the environment? Describe some skills and...
-
Is it possible for a heat engine to operate as shown in the following diagram? Explain, using the laws of thermodynamics. TH Tc QH Qc Q8 Diagram W
-
Is it possible for the efficiency of a heat engine to equal 1? Explain.
-
Since its introduction in the market in 1995, numerous mass-transfer columns have been packed with Raschig Super-Rings-a new fourth-generation packing shown in Figure 4.8-in various chemical...
-
On 1 January 2010, Elegant Fragrances Company issues 1,000,000 face value, five-year bonds with annual interest payments of 55,000 to be paid each 31 December. The market interest rate is 6.0...
-
Penben Corporation has a defined benefit pension plan. At 31 December, its pension obligation is 10 million and pension assets are 9 million. Under either IFRS or US GAAP, the reporting on the...
-
An analyst has projected that a company will have assets of 2,000 at year-end and liabilities of 1,200. The analysts projection of total owners equity should be closest to: A. 800. B. 2,000. C. 3,200.
-
Compared to a company that uses the FIFO method, during periods of rising prices a company that uses the LIFO method will most likely appear more: A. liquid. B. efficient. C. profitable. Assume the...
-
To better evaluate the solvency of a company, an analyst would most likely add to total liabilities A. the present value of future capital lease payments. B. the total amount of future operating...
-
On June 3, 2019, Alaska State Company received a $5,200, 45-day, 10 percent note from Samuel Adams, a customer whose account was past due. 1. Record in the general journal receipt of the note. 2....
-
Do the three planes x + 2x + x 3 = 4, X X 3 = 1, and x + 3x = 0 have at least one common point of intersection? Explain.
-
A 1500 kg car drives around a flat 200-m-diameter circular track at 25 m/s. What are the magnitude and direction of the net force on the car? What causes this force?
-
FIGURE Q8.5 shows two balls of equal mass moving in vertical circles. Is the tension in string A greater than, less than, or equal to the tension in string B if the balls travel over the top of the...
-
A 55 kg astronaut who weighs 180 N on a distant planet is pondering whether she can leap over a 3.5-m-wide chasm without falling in. If she leaps at a 15 angle, what initial speed does she need to...
-
Give an example of one piece of financial information that may be used differently for two stakeholders (users) and explain how it would be used and presented differently?
-
Complete common -size statements (balance sheets) using the data below on page 246. You will be required to create a spreadsheet in MS Excel to display the statements (balance sheets).? In a 1 to 2...
-
A sail boat in the Great South Bay travels 2 miles east and then 4 miles south to get to Fire Island. What is its distance and displacement?

Study smarter with the SolutionInn App


