4.0 g of oxygen gas, starting at 820°C, follow the process 1 2 shown in FIGURE P18.63....
Question:
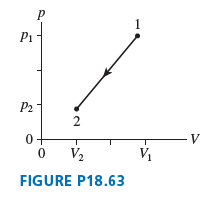
Transcribed Image Text:
P1 P2 2 0+ V2 V1 FIGURE P18.63
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (10 reviews)
Model We assume the oxygen gas is ideal Visualize From the figure w...View the full answer

Answered By

Lamya S
Highly creative, resourceful and dedicated High School Teacher with a good fluency in English (IELTS- 7.5 band scorer) and an excellent record of successful classroom presentations.
I have more than 2 years experience in tutoring students especially by using my note making strategies.
Especially adept at teaching methods of business functions and management through a positive, and flexible teaching style with the willingness to work beyond the call of duty.
Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with a colorful wing of future.
I do always believe that more than being a teacher who teaches students subjects,...i rather want to be a teacher who wants to teach students how to love learning..
Subjects i handle :
Business studies
Management studies
Operations Management
Organisational Behaviour
Change Management
Research Methodology
Strategy Management
Economics
Human Resource Management
Performance Management
Training
International Business
Business Ethics
Business Communication
Things you can expect from me :
- A clear cut answer
- A detailed conceptual way of explanation
- Simplified answer form of complex topics
- Diagrams and examples filled answers
4.90+
46+ Reviews
54+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
A 16.0-g sample of methane (CH 4 ) reacts with 64.0 g of oxygen gas in a container fitted with a piston (at 1.00 atm and 425 K). Methane can react with oxygen to form carbon dioxide and water vapor...
-
A 1.604-g sample of methane (CH4) gas and 6.400 g of oxygen gas are sealed in a 2.50- L vessel at 411oC and are allowed to reach equilibrium. Methane can react with oxygen to form gaseous carbon...
-
Exactly 4.0 g of hydrogen gas combines with 32 g of oxygen gas according to the following reaction. 2H2 + O2 2H2O a. How many hydrogen molecules are required to completely react with 48 oxygen...
-
Discuss the different sampling procedures and techniques that you will apply to the qualitative and quantitative phases of the study.
-
On May 31, 2011, Andro Corporation sold 500 shares of its stock to Nombeko, an employee, for $100 per share. No special election is made. At the time of the sale, the fair market value of the stock...
-
Find I and the power absorbed by each element in the network of Fig. 1.30. 44 A 10 A ) 15 V 15 V +. (1+
-
The water in the casing of a centrifugal pump moves (a) radially (b) axially (c) tangentially (d) in a spiral
-
Discuss why control procedures over access to the data resource become more crucial under the database approach than in the flat-file environment. What role does the DBMS play in helping to control...
-
Suppose you apply for a 7-year loan in the amount of $19,000 with an APR of 10%, compounded monthly. Your monthly payment is $315.42. Determine the total amount of interest paid over the term of the...
-
Comparative income statements and balance sheets for Coca-Cola are shown below ($ millions). Required: a. Use the following ratios to prepare a projected income statement, balance sheet, and...
-
Hasbro is a leading firm in the toy, game, and amusement industry. Its promoted brands group includes products from Playskool, Tonka, Milton Bradley, Parker Brothers, Tiger, and Wizards of the Coast....
-
10 g of dry ice (solid CO 2 ) is placed in a 10,000 cm 3 container, then all the air is quickly pumped out and the container sealed. The container is warmed to 0C, a temperature at which CO 2 is a...
-
Thad Kostowski and Carol Lee are production managers in the Appliances Division of Mesger Corporation, which has several dozen plants scattered in locations throughout the world. Carol manages the...
-
Silva Enterprises has inventory of $ 1 1 , 6 0 0 , fixed assets of $ 2 2 , 4 0 0 , total liabilities of $ 1 2 , 9 0 0 , cash of $ 1 , 9 0 0 , accounts receivable of $ 8 , 7 0 0 , and long - term debt...
-
Shruthi Singerr, a citizen and resident of country M , is a musician that puts on 5 shows on her United States tour. To minimize overhead, Shruthi manages the ticket booth before her concerts begin,...
-
The figure has permieter P=56. Find the value of x. The value of x is (Type a whole number.) +2 A x+6 (Figure is not to scale)
-
Background: XYZ Corporation, a manufacturing company, has been awarded a government grant to support its research and development activities. The grant is intended to promote innovation and...
-
The current spot rate is INR 52.888/SGD. If the Indian rupee (INR) appreciates against the Singapore dollar (SGD) by 7.56% over the coming period, then what will be the new end-of-period spot rate?...
-
Imagine that a time machine has transported you back to 1961. How was a day in the life of a salesperson selling appliances such as washing machines different in 1961 than it is now?
-
An interest bearing promissory note for 90 days at 5.6% p.a. has a face value of $120,000. If the note is discounted 20 days after the issue date at a rate of 6.8% p.a., calculate the amount of...
-
Geiger counters are not very accurate when the count rates are very high; they indicate a count rate lower than the actual value. Explain why this is so.
-
As a general rule, the radioactivity from a particular radioisotope is considered to be reduced to a safe level after 10 half-lives have elapsed. (Obviously, the initial quantity of the isotope is...
-
The naturally occurring radioisotopes uranium-238 and uranium-235 have decay chains that end with the stable isotopes lead-206 and lead-207, respectively. Natural minerals such as zircons contain...
-
x-8x+15 A) Let f(x) = = x2+2x-15 Calculate lim f(x) x 3 2x225x75 B) Let f(x) = x-18x45 Calculate lim f(x) x 15
-
If a dance instructor prices her lessons at $60 per student, she will have three students. If she prices her lessons at $50 per student, she will have four students. How much marginal revenue will...
-
What is the difference between objectives, strategies, and goals? How do these things improve our planning process?

Study smarter with the SolutionInn App


