A gas with an initial temperature of 900°C undergoes the process shown in FIGURE EX18.36. a. What
Question:
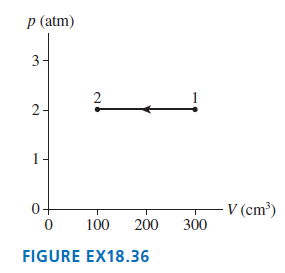 a. What type of process is this?
a. What type of process is this?
b. What is the final temperature in °C?
c. How many moles of gas are there?
Transcribed Image Text:
p (atm) 3- 2 2- 1- V (cm³) 300 0+ 100 200 FIGURE EX18.36
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
Model Assume that the gas is ideal Solve a Because the process i...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Five moles of an ideal monatomic gas with an initial temperature of 127C expand and, in the process, absorb 1200 J of heat and do 2100 J of work. What is the final temperature of the gas?
-
A 0.6-cm-thick flat plate of stainless steel, 7.5 cm wide and 0.3 m long, is immersed horizontally at an initial temperature of 980?C in a large water bath at 100?C and at atmospheric pressure....
-
A plastic tube of 7.6-cm ID and 1.27 cm wall thickness having a thermal conductivity of 1.7 W/(m K), a density of 2400 kg/m3, and a specific heat of 1675 J/(kg K) is cooled from an initial...
-
ABC Company produces mode trains. During the month of July, it produced 4,000 trains. The actual labor hours were 8 hours per train. Its standard labor hours are 10 hours per train. The standard...
-
What purpose is served by the check-the-box Regulations?
-
What is Monte Carlo simulation? What principles underlie its use, and what steps are followed in applying it?
-
When a washing machine, of mass \(200 \mathrm{~kg}\) and an unbalance \(0.02 \mathrm{~kg}\) - \(\mathrm{m}\), is mounted on an isolator, the isolator deflects by \(5 \mathrm{~mm}\) under the static...
-
Presented below are the trial balance and the other information related to Yorkis Perez, a consulting engineer. 1. Fees received in advance from clients $6,000.2. Services performed for clients that...
-
Due to shortages in the United States milk supply, Congress passed a law governing the production and sale of milk. The law required "anyone who produced, distributed, or sold milk" to obtain a...
-
In the Illustrative Case in this chapter, payroll transactions for Brookins Company were analyzed, journalized, and posted for the third quarter of the fiscal year. In this problem, you are to record...
-
0.0040 mol of gas undergoes the process shown in FIGURE EX18.35. a. What type of process is this? b. What are the initial and final temperatures in °C? p (atm) 3- 2- V (cm) 300 0+ 100 200 FIGURE...
-
0.020 mol of gas undergoes the process shown in FIGURE EX18.37. a. What type of process is this? b. What is the final temperature in °C? c. What is the final volume V 2 ? p (atm) 3- 2- 1- V (cm)...
-
Identify the typical strategy used by a long- focused AHF.
-
A large retailer wants to link two key issues into one neat solution. Employee purchases are problematic as they are irregular and unpredictable. At the same time, there is a need to overhaul the...
-
As a team, identify the roles that you think are important for teams in the small business workplace. To help you get started, here are some possible basic team roles that have been identified in...
-
Do you think that workers who complain against employers for FLSA violations may hurt themselves in the marketplace because other firms may refuse to hire them? If you were in Mr. Morimotos shoes,...
-
Suppose that you worked for an organization that promoted the establishment and running of self-managed teams. As head of HR, you have traditionally taken the lead role in the recruitment and...
-
Use the graph of f to sketch a graph of f and the graph of f. To print an enlarged copy of the graph, go to MathGraphs.com. 3 -02 1 -3 -2 -1 -3 y 1 2 3
-
What is the difference between an embedded system and a real-time system?
-
Carlton Stokes owns and operates a car-detailing business named SuperShine & Detailing. For $150, Carltons business will hand wash and wax customers cars, vacuum the interior, and thoroughly clean...
-
When a tidal barrage follows the water release profile shown in Figure 31.27 it only captures a fraction of the energy (31.35) available in the water stored at high tide. The discharge is indicated...
-
Consider an idealized tidal reservoir of surface area A = 30 km 2 that fills and drains through a narrow channel of width w = 60 m and depth d = 20 m. Assume that the tidal range is h = 2.5 m. For...
-
Estimate the total power of the Gulf Stream in the Florida Straits, given that the mass flux is roughly 30 Sv. Take the speed of the flow to be 4 m/s.
-
Recently, a famous movie star won a defamation trial against his ex-wife, also a famous movie star. The ex-husband claimed that his ex-wife had defamed him in a Washington Post column, where she...
-
On 1 August 2022, Jack, with the written approval of Mega Phones Board, then approaches Jill from Techworld to supply 500 Apple iPhones Mark XYZ, which have recently been released, for a total price...
-
Calculate Ending Inventory using Weighted average (WA) method Syd Gee Ltd Opening Trial Balance 1 June 2021 BANK STATEMENT As at 30 June 2021 DATE PARTICULARS DEBIT CREDIT BALANCE Account Debit...

Study smarter with the SolutionInn App


