A heat engine using a diatomic gas follows the cycle shown in FIGURE P21.56. Its temperature at
Question:
a. Determine Ws, Q, and ΔEthfor each of the three processes in this cycle. Display your results in a table.
b. What is the thermal efficiency of this heat engine?
c. What is the power output of the engine if it runs at 500 rpm?
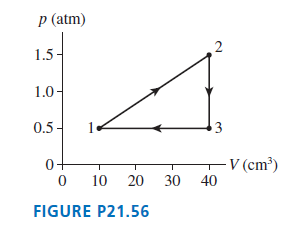
Transcribed Image Text:
p (atm) 2 1.5- 1.0- 0.5 - - V (cm³) 40 10 20 30 FIGURE P21.56 3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
Model The heat engine follows a closed cycle For a diatomic gas C V 52 R and C P 72 R Solve ...View the full answer

Answered By

Deepak Pal
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students. Areas of interest: Business, accounting, Project management, sociology, technology, computers, English, linguistics, media, philosophy, political science, statistics, data science, Excel, psychology, art, history, health education, gender studies, cultural studies, ethics, religion. I am also decent with math(s) & Programming. If you have a project you think I can take on, please feel welcome to invite me, and I'm going to check it out!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
A heat engine using a diatomic ideal gas goes through the following closed cycle: Isothermal compression until the volume is halved. Isobaric expansion until the volume is restored to its initial...
-
A heat engine uses a diatomic gas in a Brayton cycle. What is the engines thermal efficiency if the gas volume is halved during the adiabatic compression?
-
Consider a heat engine that is not reversible. The engine uses 1.000 mol of a diatomic ideal gas. In the first step (A) there is a constant temperature expansion while in contact with a warm...
-
2. LIMIT COMPARISON TEST: SPECIAL CASES The Limit Comparison Test is most often used when the limit L= lim ak lies in the interval (0, oo). In the case where L is zero or infinity, we can still...
-
Evaluate the competitive strategies for firms in foreign markets: a. Niche market exporting b. Licensing and contract manufacturing c. Franchising d. Joint ventures e. Wholly owned subsidiaries
-
Fill in the blanks with an appropriate word, phrase, or symbol(s). The number 1/2 can be represented as a(n) ___________ decimal number.
-
Define true and engineering stresses.
-
Primera Banco is evaluating two capital investment proposals for a drive-up ATM kiosk, each requiring an investment of $360,000 and each with an 8-year life and expected total net cash flows of...
-
Atari, founded in 1972, launched the video game industry with its big hit, Pong . As Atari grew during its first 5 years, Atari co-founder and avid Disney fan Nolan Bushnell recognized the value of...
-
Read the case study "Google: new product innovation at the speed of light" and answer the following questions: 1. Identify major similarities and differences between the new products development...
-
A heat engine using 1.0 mol of a monatomic gas follows the cycle shown in FIGURE P21.55. 3750 J of heat energy is transferred to the gas during process 1 2. a. Determine W s , Q, and ÎE th for...
-
FIGURE P21.57 shows the cycle for a heat engine that uses a gas having Ï = 1.25. The initial temperature is T 1 = 300 K, and this engine operates at 20 cycles per second. a. What is the power...
-
What is Tobins q, and how does it link financial markets to capital investment?
-
Using the Kraljic model, analyse how sustainability can be achieved in supply activities. In light to manufacturing activities, what are some of the sustainable practices that can be adopted in a)...
-
Discuss and explain the effectiveness of the use of price as quality cue by marketers. Instructions: Introduction Body Conclusion Please add in text citations and very good, relevant and recent...
-
Radom Manufacturing produces various products. The company operates a landfill, which it uses to dispose of nonhazardous trash. The trash is hauled from the two nearby manufacturing facilities in...
-
At today's executive meeting your company's CEO said: "On the planning and strategic level, we need holistic marketing problem solvers who are just as likely to recommend a product improvement as...
-
By how much does the fluid level rise in the side of the manometer that is open to the atmosphere? By how much does the fluid level rise in the side of the manometer that is open to the atmosphere if...
-
Explain the differences between a fixed WiMAX and a mobile WiMAX.
-
Write electron configurations for the following ions, and determine which have noble-gas configurations: (a) Cd2+ (b) p3- (c) Zr4+ (d) Ru3+ (e) As3- (f) Ag+
-
Two masses are separated by a distance r. If this distance is doubled, is the force of interaction between the two masses doubled, halved, or changed by some other amount? Explain.
-
A painter depicts a portion of the night sky as shown in the diagram below, showing the stars and a crescent moon. Is this view possible? Explain. Q24 Diagram
-
At what times during the day or night would you expect the new moon to rise and set? Explain.
-
Explain possible advantages and disadvantages of PSL's current international trading strategy (subsidiary in China, export & import and online presence).
-
In November 2020 you entered into four May 2021 long futures contracts for crude oil (1,000 barrels per contract) for $65.00 per barrel. If the futures price was $70.00 per barrel on December 31,...
-
4) Champagne, Inc., had revenues of $13 million, cash operating expenses of $9 million, and depreciation and amortization of $2 million during 2008. The firm purchased $650,000 of equipment during...

Study smarter with the SolutionInn App


