Draw an energy-level diagram, similar to Figure 38.21 , for the He + ion. On your diagram:
Question:
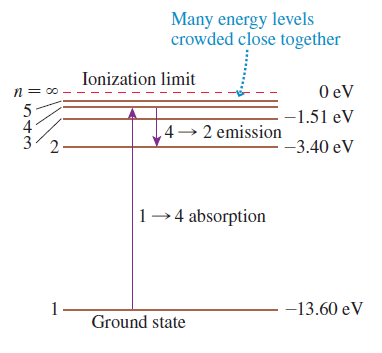 a. Show the first five energy levels. Label each with the values of n and En.
a. Show the first five energy levels. Label each with the values of n and En.
b. Show the ionization limit.
c. Show all possible emission transitions from the n = 4 energy level.
d. Calculate the wavelengths (in nm) for each of the transitions in part c and show them alongside the appropriate arrow.
Transcribed Image Text:
Many energy levels crowded close together Ionization limit O eV -1.51 eV 4 4→ 2 emission -3.40 eV 2. 1-4 absorption -13.60 eV Ground state
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
Visualize Solve a He is a hydrogenlike ion with Z 2 From Equation 3841 its energy levels ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
A particle confined in a rigid one-dimensional box of length 10 fm has an energy level E n = 32.9 MeV and an adjacent energy level E n+1 = 51.4 MeV. a. Determine the values of n and n + 1. b. Draw an...
-
The following is an energy- level diagram illustrating three different electronic transitions in the Bohr hydrogen atom. a. Explain why the energy levels get closer together as they increase. Provide...
-
An atom has a line spectrum consisting of a red line and a blue line. Assume that each line corresponds to a transition between two adjacent energy levels. Sketch an energy-level diagram with three...
-
Suggest a systematic process that Daimler could have followed to increase the likelihood of merger success. Provide examples on how Daimler might have made such a process succeed.
-
Three parachutists, A, B, and C, each have reached terminal velocity at the same distance above the ground below. a. From fastest to slowest, rank their terminal velocities. b. From longest to...
-
It is the end of 2013. The All-Fixed Company began operations in January 2012. The company is so named because it has no variable costs. All its costs are fixed; they do not vary with output. The...
-
What is the difference between bits that are used in todays computers and qubits that are used in quantum computers?
-
A machine costing $257,500 with a four-year life and an estimated $20,000 salvage value is installed in Luther Companys factory on January 1. The factory manager estimates the machine will produce...
-
Consider a flywheel that is a solid cylindrical disk 0.2 meters in radius with a mass of 40 kg. The flywheel turns at 700 rpm until acceleration is applied by an external torque; in 6 seconds the...
-
Jimmy owns a garden in which he has planted N trees in a row. After a few years, the trees have grown up and now they have different heights. Jimmy pays much attention to the aesthetics of his...
-
The muon is a subatomic particle with the same charge as an electron but with a mass that is 207 times greater: m = 207m e . Physicists think of muons as heavy electrons. However, the muon is not a...
-
Consider a hydrogen atom in stationary state n. a. Show that the orbital period of an electron in quantum state n is T = n 3 T 1 , and find a numerical value for T 1 . b. On average, an atom stays in...
-
Check that Equation 11.103 satisfies the time-dependent Schrdinger equation for the Hamiltonian in Equation 11.97. Also confirm Equation 11.105, and show that the sum of the squares of the...
-
Use the following transactions to complete the second month of operations for Billy Bob's Daycare. Feb. Billy Bob began selling T-shirts to the kids and parents. Billy pays cash for 40 T- shirts at...
-
John Conway was a brilliant mathematician who spent much of his career at Princeton University. One of his (many) contributions to was to create a "game" called Life. In reality though, it isn't a...
-
Using your current cost structure and earring price assumptions, create a CVP graph showing cost and revenue data from zero to 750,000 earrings. Clearly indicate the break-even point on the graph....
-
You need to raise investment capital and decide the best method is selling securities (stock'shares of ownership interests) in the corporation you formed by converting from an LLC. Before you do...
-
4 You are a tech start-up entrepreneur. With a passion to build the next unicorn from India, you are expected to develop and present a detailed assignment as a part of the comprehensive Business...
-
Calculate the mass of a can of oil if it weighs 610 N.
-
The following information is available for Partin Company: Sales $598,000 Sales Returns and Allowances 20,000 Cost of Goods Sold 398,000 Selling Expense 69,000 Administrative Expense 25,000 Interest...
-
In thermal management or cooling of microchip modules for high-powered computer systems, very high heat fluxes have to be accommodated in the design of the cooling method. For many such cases,...
-
Lubricating oil is cooled in a tubular heat exchanger to maintain its viscosity and effectiveness in the journal bearings used in a large steam turbine of an electric power plant. Oil flows at the...
-
Purified water, flowing at the rate of 45 kg/h is to be heated from 25C to 75C in a food processing plant before it is mixed with thickened tomato puree for sauce production. A thin-walled, 1.25 cm...
-
ABC Hospital is one of two hospitals in the community. The Hospital offers all major specialty services (e.g. cardiology, neurology, pulmonology, etc.) The Hospital is approached by a cardiologist...
-
You obtained job-related information about the HR assistant position from O*Net. Answer the following sections based on that information. QUESTIONS: The following is the list of questions included in...
-
On January 8, the end of the first weekly pay period of the year, Regis Company's employees earned $27,760 of office salaries and $70,840 of sales salaries. Withholdings from the employees' salaries...

Study smarter with the SolutionInn App


