Figure CP19.79 shows a thermodynamic process followed by 0.015 mol of hydrogen. How much heat energy is
Question:
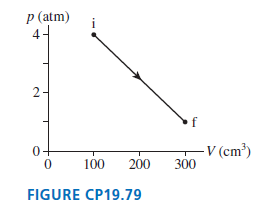
Transcribed Image Text:
p (atm) 2- -V (cm³) 300 100 200 FIGURE CP19.79
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Assume the gas to be an ideal gas Solve The work done ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Figure P19.62 shows a thermodynamic process followed by 120 mg of helium. a. Determine the pressure (in atm), temperature (in C), and volume (in cm 3 ) of the gas at points 1, 2, and 3. Put your...
-
Figure Q16.10 shows a thermodynamic process in which a system first expands and is then compressed. Indicate the area on the PV diagram that corresponds to the work done by the system. Figure Q16.10...
-
A Thermodynamic Process in an Insect the African bombardier beetle Stenaptinus insignis can emit a jet of defensive spray from the movable tip of its abdomen (Fig. 19.32). The beetle's body has...
-
Reid Corporation's balance sheet at January 1, 20X9 reflected the following balances: Cash & Receivables $ 30,000 Inventory $ 75,000 Land $125,00 Building & Equipment (net) $850,000 Common Stock...
-
Glen and Diane Okumura (both age 48) are married, file a joint return, and live at 39 Kaloa Street, Honolulu, HI 96815. Glen's Social Security number is 111-11-1111, and Diane's is 123-45-6789. The...
-
Andreas is working on estimates using features. In which step would he be working with the product manager on this?
-
In a vapour compression refrigeration system, the condition of refrigerant before entering the compressor is (a) saturated liquid (b) wet vapour (c) dry saturated liquid (d) superheated vapour
-
It is now January. The current interest rate is 5%. The June futures price for gold is $946.30, whereas the December futures price is $960.00. Is there an arbitrage opportunity here? If so, how would...
-
Six months after the triple homicide patrol officers get a call of a suspicious subject at 3:00 am. The complainant observes the suspect walking down the street and disappear behind a neighbors...
-
Enterprises began business some years ago as a franchiser of fast food restaurants known as The Mighty Chicken. Initially, all franchises were independently owned, but eventually the company began to...
-
A beaker with a metal bottom is filled with 20 g of water at 20C. It is brought into good thermal contact with a 4000 cm 3 container holding 0.40 mol of a monatomic gas at 10 atm pressure. Both...
-
One cylinder in the diesel engine of a truck has an initial volume of 600 cm 3 . Air is admitted to the cylinder at 30C and a pressure of 1.0 atm. The piston rod then does 400 J of work to rapidly...
-
What are convertible bonds and exchangeable bonds?
-
9 months ago, you went long 3 Lithium future contracts at a price of $481705 per 1000 kg. You put down a margin of 10% and are about to go short the same number of contracts but at a price of...
-
ESSAY. Write your answer in the space provided e Points: 15 1) Describe the working methodology for the followings: 1. List 2. Queue Try to describe the elemtent positions, inertion critieria,...
-
The federal minimum wage and 40-hour workweek were firstintroduced in 1938 by Congress under the Fair Labor Standards Act(FLSA). FDR, president at the time, claimed the law was the mostfar-sighted...
-
Business people encounter many legal issues beyond the topics covered in a typical Business Law course. This report gives you the opportunity to explore an additional legal topic that has potential...
-
Find the value of if sin x = y, then (A) 0 y (B) (c) 0 < y < (D) 22
-
A sunglasses retailer is trying to determine if there is a significant market for its merchandise in a specific mall location where it is considering opening a store. It has an active Facebook page...
-
From 1970 to 1990, Sri Lanka's population grew by approximately 2.2 million persons every five years. The population in 1970 was 12.2 million people.What is the best formula for P, Sri Lanka's...
-
As depicted in Figure 12.19, are gallium atoms really red and arsenic atoms green? Figure 12.19 (a) (c) (b)
-
With scanning probe microscopy technology, we see not actual atoms, but rather images of them. Explain.
-
Why is it not possible for a scanning probe microscope to make images of the inside of an atom?
-
There are two real numbers a for which (2x)+15 (1-x) = 8x - 3x + 2. The sum of these two real numbers is
-
Net sales Cost of goods sold Depreciation INCOME STATEMENT (Figures in $ millions) Earnings before interest and taxes (EBIT) Interest expense Income before tax Taxes Net income $ 882.00 742.00 32.00...
-
Venita is sewing new curtains. The table below shows the relationship between the number of stitches in a row and t length of the row. Sewing Number of Length (cm) stitches 5 1 15 3 20 4 30 6 What is...

Study smarter with the SolutionInn App


