Figure P19.62 shows a thermodynamic process followed by 120 mg of helium. a. Determine the pressure (in
Question:
Figure P19.62 shows a thermodynamic process followed by 120 mg of helium.
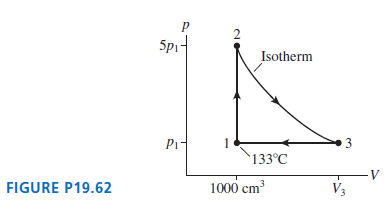 a. Determine the pressure (in atm), temperature (in °C), and volume (in cm3) of the gas at points 1, 2, and 3. Put your results in a table for easy reading.b. How much work is done on the gas during each of the three segments?c. How much heat energy is transferred to or from the gas during each of the three segments?
a. Determine the pressure (in atm), temperature (in °C), and volume (in cm3) of the gas at points 1, 2, and 3. Put your results in a table for easy reading.b. How much work is done on the gas during each of the three segments?c. How much heat energy is transferred to or from the gas during each of the three segments?
Transcribed Image Text:
2 5pi- Isotherm `133°C V FIGURE P19.62 1000 cm V3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
Model Assume that the gas is an ideal gas and that the work heat and thermal ene...View the full answer

Answered By

Umber Talat
I am providing full time mentoring and tutoring services in Business Finance, Contemporary issue in Global Economy, Quantitative Techniques, Principles of Marketing, strategic marketing, International Marketing, Organizational Behavior (OB), Consumer Behavior, Sales Force Management, Strategic Brand Management, Services Marketing, Integrated Marketing Communication (IMC), Principles of Management, General Management, Strategic Management, Small and Medium Enterprise Management, Innovation Management, Change Management, Knowledge Management, Strategic Planning, Operations Management, Supply Chain Management, Logistics Management, Inventory management, Total Quality Management (TQM), Productions Management, Project Management, Production Planning, Human Resource Management (HRM), Human Resource Development, Strategic HRM, Organizational Planning, Performance and Compensation Management, Recruitment and Selection, Organizational Development, Global Issues in Human Resource Management, Retail Marketing, Entrepreneurship, Entrepreneurial Marketing, International Business, Research Methods in Business, Business Communication, Business Ethics.
4.70+
158+ Reviews
236+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Figure CP19.79 shows a thermodynamic process followed by 0.015 mol of hydrogen. How much heat energy is transferred to the gas? p (atm) 2- -V (cm) 300 100 200 FIGURE CP19.79
-
Figure Q16.10 shows a thermodynamic process in which a system first expands and is then compressed. Indicate the area on the PV diagram that corresponds to the work done by the system. Figure Q16.10...
-
A Thermodynamic Process in an Insect the African bombardier beetle Stenaptinus insignis can emit a jet of defensive spray from the movable tip of its abdomen (Fig. 19.32). The beetle's body has...
-
Find the deflection y(x) of a cantilever beam embedded at its left end and free at its right end when the load is as given in Example 10.
-
List the quantum numbers for each electron in the ground state of oxygen (Z = 8).
-
Fallon Company, a toy manufacturer that also operates several retail outlets, is preparing its December 31, 2007 financial statements. It has identified the following legal situations that may...
-
What are your sales objectives? What extra information about Mr Forbes would be useful to have?
-
The following transactions of Kelsey, Inc., occurred within the same accounting period: (a) Purchased $55,000 U.S. Treasury 6% bonds, paying 102 plus accrued interest of $1,400. In addition, Kelsey...
-
If the intensity of radiation incident on a surface is uniform in all directions and denoted by the constant I, verify that the total flux is I. Note that this approximately describes the
-
The time-series graphs below show typical simulation output. For each graph identify the type of model (terminating or nonterminating) and the nature of the simulation output (transient,...
-
Two cylinders each contain 0.10 mol of a diatomic gas at 300 K and a pressure of 3.0 atm. Cylinder A expands isothermally and cylinder B expands adiabatically until the pressure of each is 1.0 atm....
-
Two containers of a diatomic gas have the same initial conditions. One container, heated at constant pressure, has a temperature increase of 20C. The other container receives the same quantity of...
-
Follow the directions of Problem 17, but revolve R about the line y = 4. In problem 17 Find the volume of the solid generated by revolving the region R bounded by the curves x = y and x = y3/32 about...
-
State University needs a listing of the overall SAT percentiles of the 14,226 students it has accepted in the past year. The data are in a text file, with one line per student. That line contains the...
-
The specifications for the Stack ADT have been changed. The class representing the stack must now check for overflow and underflow and set an error flag (a parameter) to true if either occurs. 1....
-
Store the values in a hash table with 20 positions, using rehashing as the method of collision resolution. Use key % tableSize as the hash function, and (key + 3) % tableSize as the rehash function....
-
The article "Two Different Approaches for RDC Modelling When Simulating a Solvent Deasphalting Plant" (J. Aparicio, M. Heronimo, et al., Computers and Chemical Engineering, 2002:1369-1377) reports...
-
Show what is written by the following segments of code, given that item1, item2, and item3 are int variables. 1. StackType stack; item1 = 1; item2 = 0; item3 = 4; stack.Push(item2);...
-
Plaintiff filed a petition for preliminary injunction asking the court to enjoin the sale of a 1991 Ferrari F40 to Karam Automotive Inc., as well as a complaint seeking specific performance of the...
-
Which of the following statements is false? a. Capital leases are not commonly reported in a Capital Projects Fund. b. A governmental entity may report a Capital Project Fund in one year but not the...
-
How many mirrors produce these multiple reflections of physics teacher Karen Jo Matsler?
-
Below is a photo of science author Suzanne Lyons with her son Tristan wearing red and her daughter Simone wearing green. Below that is the negative of the photo, which shows these colors differently....
-
Sunlight passing through a pinhole in a piece of paper casts an image of the Sun, as shown. The image size depends on the distance from the pinhole to the floor. If the paper with the pinhole is held...
-
A regional electrical distributor currently has 1,000 customers who buy an average of $5,000 per year, generating a 50% margin. From experience, the company knows that 20% of its customers will not...
-
Suppose you observed that one-year T-bills are trading with a yield to maturity (YTM) of 4.75%. The yield spread between AAA and BB rated corporate bonds is 130 basis points. The maturity yield...
-
You have $36,000 to Invest in Sophie Shoes, a stock selling for $60 a share. The initial margin requirement is 65 percent. calculate your rates of return if the stock rises to $90 a share and if it...

Study smarter with the SolutionInn App


