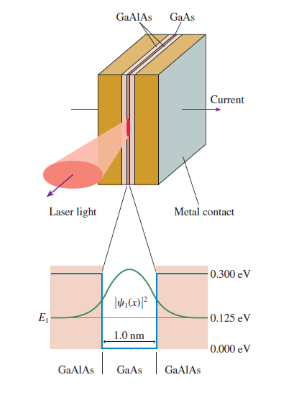
For the quantum-well laser of Figure 40.16 , estimate the probability that an electron will be found
Question:
Transcribed Image Text:
GAAIAS GạAs Current Laser light Metal contact -0.300 eV 0.125 eV 1.0 nm 0.000 eV |GAAIAS GAAIAS GaAs
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Solve Finding the electron in the GaAlAs is equivalent to finding the electron ...View the full answer

Answered By

Subash Murugaih
I am leading expert in this web site couple of years and My clients are much happy with my works and services.
4.60+
309+ Reviews
539+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
FIGURE P39.32 shows |(x)| 2 for the electrons in an experiment. a. Is the electron wave function normalized? Explain.b. Draw a graph of (x) over this same interval. Provide a numerical scale on both...
-
Even the smoothest mirror finishes are rough when viewed at a scale of 100 nm. When two very smooth metals are placed in contact with each other, the actual distance between the surfaces varies from...
-
What is the probability that an electron will jump across the energy gap Eg in a diamond that has a mass equal to the mass of Earth? Use the result of Sample Problem 41-1 and the molar mass of carbon...
-
Payroll Assignment - (50 Marks) The following employees are working in the ABC Clinic, they are paid biweekly. Calculate the gross income, net income, and the total remittance that ABC is to report...
-
Which interact more with high-frequency light: small particles or large particles?
-
On June 30, 2014, the end of the first month of operations, Tudor Manufacturing Co. prepared the following income statement, based on the variable costing concept: a. Prepare an absorption costing...
-
Under Statement on Auditing Standards No. 57, "Auditing Accounting Estimates," an auditor is responsible for: a. Making accounting estimates. b. Evaluating the reasonableness of management's...
-
Single-rate versus dual-rate methods, support department. The Chicago power plant that services all manufacturing departments of MidWest Engineering has a budget for the coming year. This budget been...
-
(The Sieve of Eratosthenes) A prime integer is any integer greater than 1 that can be divided evenly only by itself and 1. The Sieve of Eratosthenes is a method of finding prime numbers. 1. Create an...
-
A quarterback on a football team has a pass completion rate of 0.62. If, in a given game, he attempts 16 passes, what is the probability that he will complete (a) 12 passes? (b) More than half of his...
-
A neutron is confined in a 10-fm-diameter nucleus. If the nucleus is modeled as a one-dimensional rigid box, what is the probability that a neutron in the ground state is less than 2.0 fm from the...
-
For a particle in a finite potential well of width L and depth U 0 , what is the ratio of the probability Prob(in x at x = L + ) to the probability Prob(in x at x = L)?
-
Describe a primary key, candidate key, secondary key, foreign key, and a combination key. Use your imagination to provide an example of each key that is not in the textbook.
-
The following information is available from the 2024 financial statements of Indigo Inc. and Sandhill Ltd.: Income tax expense Interest expense Net income Total assets Total current liabilities Total...
-
ONE require an adjustment (Adjusting Entry AJE) a) Prepaid Insurance b) Salary Expense - c) Interest Payable - d) Service Fees Earned - e) Unearned Revenue - f) Depreciation Expense - 4. Emery...
-
Course Hero Ch 03 Homework 2 2 points Skipped eBook 01 Hint Print References Mc Graw Hill Revenues Services revenue Commissions revenue Total revenues Expenses Exercise 3-15 (Algo) Analyzing and...
-
Look up the brand valuation of IKEA using the three prominent brand valuation sources and explain why the results varied between them. 1) Brand Z -...
-
(b) Answer BOTH parts (i) and (ii). (i) Suggest a structure for A and provide a mechanism for its formation. i). NaOEt/EtOH ii). CH3-1 A 6H8O2 NaOD/DO (ii) Suggest a structure for B and account for...
-
The circular shift operation is one of the components of the modern block ciphers. a. Show the result of a 3-bit circular left shift on the word (10011011) 2 . b. Show the result of a 3-bit circular...
-
Pedro Bourbone is the founder and owner of a highly successful small business and, over the past several years, has accumulated a significant amount of personal wealth. His portfolio of stocks and...
-
A beaker with a metal bottom is filled with 20 g of water at 20C. It is brought into good thermal contact with a 4000 cm 3 container holding 0.40 mol of a monatomic gas at 10 atm pressure. Both...
-
Figure CP19.79 shows a thermodynamic process followed by 0.015 mol of hydrogen. How much heat energy is transferred to the gas? p (atm) 2- -V (cm) 300 100 200 FIGURE CP19.79
-
One cylinder in the diesel engine of a truck has an initial volume of 600 cm 3 . Air is admitted to the cylinder at 30C and a pressure of 1.0 atm. The piston rod then does 400 J of work to rapidly...
-
Mrs. Sam Taffer was a brilliant engineer at IBM and made a lot of money from stock options. Since then, she has worked as a musician and raised her two sons. She wishes to have most of her property...
-
Miller Company ended its fiscal year on June 30, 2017. The company's adjusted trial balance as of the end of its fiscal year is shown below. MILLER COMPANY Adjusted Trial Balance June 30, 2017...
-
What depreciation method is used to write off property, plant and equipment? Regarding the triple bottom line, what information did the company disclose about environmental matters in the reports?...

Study smarter with the SolutionInn App


