The two springs in FIGURE P29.72 each have a spring constant of 10 N/m. They are compressed
Question:
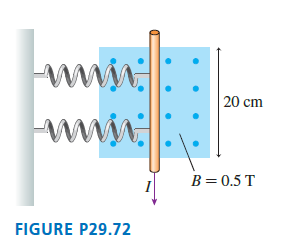
Transcribed Image Text:
www 20 cm www. B=0.5 T FIGURE P29.72
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
Model A magnetic field exerts a magnetic force on a length of currentcarrying wire We igno...View the full answer

Answered By

Antony Mutonga
I am a professional educator and writer with exceptional skills in assisting bloggers and other specializations that necessitate a fantastic writer. One of the most significant parts of being the best is that I have provided excellent service to a large number of clients. With my exceptional abilities, I have amassed a large number of references, allowing me to continue working as a respected and admired writer. As a skilled content writer, I am also a reputable IT writer with the necessary talents to turn papers into exceptional results.
4.50+
2+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
The spring in FIGURE EX10.22a is compressed by Îx. It launches the block across a frictionless surface with speed v 0 . The two springs in FIGURE EX10.22b are identical to the spring of Figure...
-
A 10.0-cm cylindrical chamber has a 5.00-cm-diameter piston attached to one end. The piston is connected to an ideal spring with a spring constant of 10.0 N/cm, as shown. Initially, the spring is not...
-
The spring in FIGURE CP10.73 has a spring constant of 1000 N/m. It is compressed 15 cm, then launches a 200 g block. The horizontal surface is frictionless, but the blocks coefficient of kinetic...
-
Consider the following differential equation describing a plug flow bioreactor: dy --Dy+py dt dz -(4-2)D-2uy dt where D = 0.4, 0.52/(0.1+),and the initial conditions are y(0) = 1 and z(0) = 4. a)...
-
An electron and a proton have the same de Broglie wavelength. (a) Which has the greater kinetic energy? Explain. (b) Calculate the ratio of the electron's kinetic energy to the kinetic energy of the...
-
What's the present value of $1,000 to be received in 8 years? (Your required rate of return is 7% a year.)
-
This Monte Carlo experiment demonstrates the inconsistency of the ordinary least squares estimator and the efficacy of instrumental variables estimation for the errors in variables problem. Consider...
-
The Poster Bed Company believes that its industry can best be classified as monopolistically competitive. An analysis of the demand for its canopy bed has resulted in the following estimated demand...
-
Your company has an on-premises Active Directory domain and a new Microsoft 365 E5 subscription. You plan to sync on-premises Active Directory objects to Microsoft Entra. You need to identify which...
-
1. What is the demographic and rock climbing profile of Extreme Exposure's members? 2. How satisfied are the members with Extreme Exposure's climbing facilities? 3. How interested are its members in...
-
The 10-turn loop of wire shown in FIGURE P29.71 lies in a horizontal plane, parallel to a uniform horizontal magnetic field, and carries a 2.0 A current. The loop is free to rotate about a...
-
FIGURE P29.73 is an edge view of a 2.0 kg square loop, 2.5 m on each side, with its lower edge resting on a frictionless, horizontal surface. A 25 A current is flowing around the loop in the...
-
Showthat the metal centres in the following complexes obey the 18-electron rule: (a) (n-Cp)Rh(n-CH4) (PMe3) (b) (n-C3H5)2 Rh (-Cl) Rh(n-C3H5) 2 (c) Cr(CO)4 (PPH3)2 (d) Fe(CO)3 (n-CHCHCHCH) (e) Fe...
-
Investigate the formation of stalactites and stalagmites. Dissolve the largest quantity of Epsom salt you can in a jar full of water. Fill two small cups with the solution, set them on paper towels,...
-
According to the graph, what is average atmospheric pressure at sea level? Why does atmospheric pressure change with altitude as shown in this graph? Altitude (km) 40 36 32 28 24 22 20 16 12 8 4 0...
-
Because John was so happy that Dana had agreed to become his fiancee, on February 2, 2018, he gave Dana's mother, Ethel, \(\$ 15,000\) in cash. a. Does this gift qualify for gift-splitting? b. Does...
-
In 2018, Mr. Colburn created a trust for the benefit of his five adult children and transferred \(\$ 300,000\) to it. The trustee may decide how much income to pay to each child each year. The trust...
-
During 2018, Alec Meachin earned a net profit of \(\$ 80,000\) from his sole proprietorship (as reported on Schedule C). He paid \(\$ 10,000\) self-employment taxes during the year. What is the...
-
How is yield to maturity related to the concept of rate of return?
-
For Problem estimate the change in y for the given change in x. y = f(x), f'(12) = 30, x increases from 12 to 12.2
-
Given the conditions of Problem 21.16, what height of vertical wall will cause the film at the bottom of the tube to be turbulent?
-
A vertical flat surface 2 ft high is maintained at 60F. If saturated ammonia at 85F is adjacent to the surface, what heat- transfer coefficient will apply to the condensation process? What total heat...
-
A single tube-pass heat exchanger is to be designed to heat water by condensing steam in the shell. The water is to pass through the smooth horizontal tubes in turbulent flow, and the steam is to be...
-
Give an example of one piece of financial information that may be used differently for two stakeholders (users) and explain how it would be used and presented differently?
-
Complete common -size statements (balance sheets) using the data below on page 246. You will be required to create a spreadsheet in MS Excel to display the statements (balance sheets).? In a 1 to 2...
-
A sail boat in the Great South Bay travels 2 miles east and then 4 miles south to get to Fire Island. What is its distance and displacement?

Study smarter with the SolutionInn App


