According to Figure 2, which of the following compounds would most typically be found only in rocks
Question:
According to Figure 2, which of the following compounds would most typically be found only in rocks at the moderate stage?
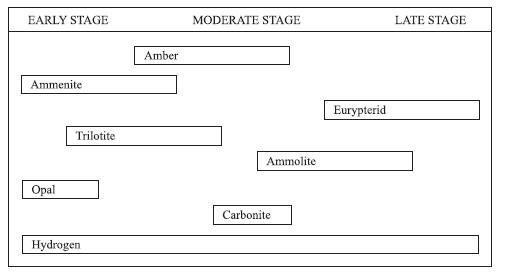
F. Ammoenite
G. Eurypterid
H. Carbonite
J. Clay
Transcribed Image Text:
EARLY STAGE Ammenite Opal Trilotite Hydrogen Amber MODERATE STAGE Ammolite Carbonite Eurypterid LATE STAGE
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The best answer is H According to Figure 2 the only compound that i...View the full answer

Answered By

Anjali Arora
Having the experience of 16 years in providing the best solutions with a proven track record of technical contribution and appreciated for leadership in enhancing team productivity, deliverable quality, and customer satisfaction. Expertise in providing the solution in Computer Science, Management, Accounting, English, Statistics, and Maths.
Also, do website designing and Programming.
Having 7 yrs of Project Management experience.
100% satisfactory answers.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Business questions
-
1. In the classical model, it is thought that the long-run: A. and short-run aggregate supply curves are both upward sloping. B. aggregate supply curve is vertical and the short-run aggregate supply...
-
Which of the following compounds would you expect to be the most generally reactive, and why? DO
-
Which of the following compounds would you expect to exhibit only London forces? a. Potassium chloride, KCl b. c. Silicon tetrafluoride, SiF4 d. Phosphorus pentachloride, PCl5 (
-
Jillian operates a sporting goods store in a rented location at a shopping mall. She is insured under a CGL policy with the following limits: Indicate the dollar amount, if any, that Jillians insurer...
-
Plattsburg Plastics Corporation manufactures a variety of plastic products, including a series of molded chairs. The three models of molded chairs, which are all variations of the same design, are...
-
A single-pass cross-flow heat exchanger uses hot air (mixed) to heat water (unmixed), flowing with a mass flow rate of 3 kg/s, from 30°C to 80°C. The hot air enters and exits the heat...
-
Assuming the same data as given in problem 9, was the well in each case profitable? Discuss your answer. Problem 9:- Property cost (acquisition cost). Drilling cost (one well). Estimated completion...
-
The marketing department of Metroline Manufacturing estimates that its sales in 2013 will be $1.5 million. Interest expense is expected to remain unchanged at $35,000, and the firm plans to pay...
-
A 2.00 m rod is rotating with an oscillating angular momentum given by sin(2.00) . If a small rocket with oscillating linear momentum 10.0cos(2.00) is fired at =0.00 s , at what time will it need to...
-
Cast fossils are formed when material is placed into a depression in a rock near the surface. They are formed at very high temperatures and low pressures. According to Figure 1, cast fossils are most...
-
Which of the following graphs best represents the relationship between time elapsed and saturation of the rocks observed in Experiment 1? Experiment 1 In a laboratory, a scientist wanted to determine...
-
For the problem presented in Examples 10.1 and 10.2, determine the discounted cash flow rate of return (DCFROR). Example 10.2 For the project described in Example 10.1, determine the following...
-
Hal is single and in 2018 he obtained IRS permission to change from the calendar year to a fiscal year ended August 31, 2018. Hal earned \(\$ 80,000\) through August 31, 2018 and had \$7,000 in...
-
How does the use of a multiple regression model affect the number of degrees of freedom in a regression model? Contrast your response with that involving a simple regression model.
-
Ruben and Albert are both calendar-year taxpayers, and are equal partners in the RA Partnership, which has a fiscal year ending on October 31. During the 2018-2019 fiscal year the partnership earns...
-
The Light Record Corporation has been on the calendar year since its inception five years ago. It wishes to change to an April 30 natural business year. For 2018, the calendar year of proposed...
-
Describe several examples of the impact that entrepreneurial firms have on a society.
-
By which method, cost or equity, do IFRSs require a parent company to record its investment in a subsidiary? Why?
-
Use the T account for Cash below to record the portion of each of the following transactions, if any that affect cash. How do these transactions affect the companys liquidity? Jan. 2 Provided...
-
If the demand for perfume is inelastic above and below the present price, should the price be raised? Why or why not?
-
If the demand for shrimp is highly elastic below the present price, should the price be lowered?
-
Discuss what factors lead to inelastic demand and supply curves. Are they likely to be found together in the same situation?
-
calculate the moving average cost from this data.... January 15 Purchase 55 17 January 16 Purchase return 5 17 January 20 Sale 91 31 January 25 Purchase 17 19
-
CAMERON & JOCELYN (married) Cameron and Jocelyn have 3 children, aged 4, 13, and 17, and also care for Jocelyn's homebound mother. They live and work in Las Vegas and take full advantage of all the...
-
How do I produce a T account when someone has paid business rent from his personal bank account? I'm assuming this would be recorded as a debit in the rent account but I'm not sure about the bank...

Study smarter with the SolutionInn App


