Consider the separation of inorganic and organic anions in Figure 26-7. a. What is the probable charge
Question:
Consider the separation of inorganic and organic anions in Figure 26-7.
a. What is the probable charge (Xn-) (Xn-)of pyruvate (peak 1010), 2-2-oxovalerate (peak 1616), and maleate (peak 2828)?
b. Iodide (peak 44 44) is a -1-1 ion. Explain its strong retention.
Figure 26-7
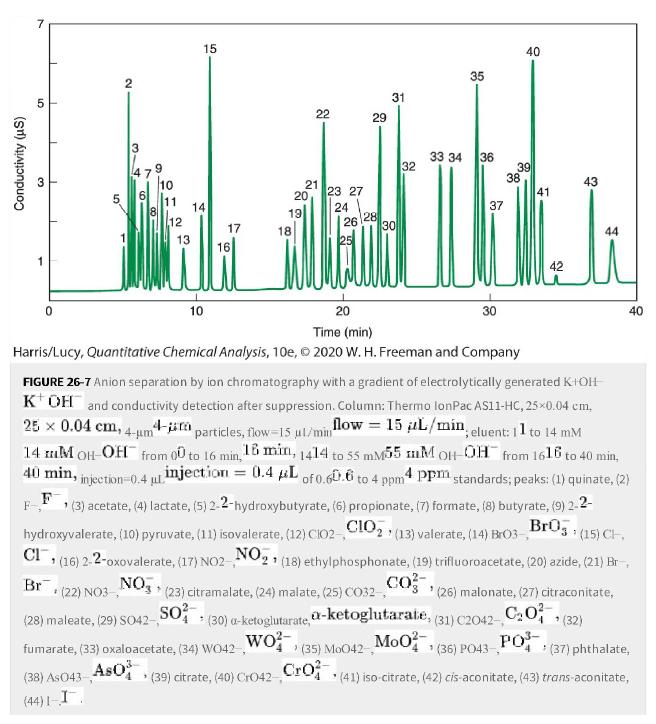
Transcribed Image Text:
Conductivity (us) 31 3 T 50 2 3 A 6 9 10 11 15 12 17 ili 10 19 18 20 22 21 23 27 24 26 29 28 31 . (39) citrate, (40) Cro42 Cro. 30 32 (35) Mo042- 33 34 35 36 37 1 30 20 Time (min) Harris/Lucy, Quantitative Chemical Analysis, 10e, © 2020 W. H. Freeman and Company 39 38 40 41 43 44 FIGURE 26-7 Anion separation by ion chromatography with a gradient of electrolytically generated K+OH KOH and conductivity detection after suppression. Column: Thermo lonPac AS11-HC, 25×0.04 cm, 25 x 0.04 cm, 4-um4 4- particles, flow-15 µl/min flow = 15 L/min, eluent: 11 to 14 mM 14 M OH-OH from 0 to 16 min, 18 min, 1414 to 55 mM55 mM OH-OH from 1616 to 40 min, 40 min, injection 0.4 injection = 0.4 μL of 0.60.6 to 4 ppm4 ppm standards; peaks: (1) quinate, (2) (3) acetate, (4) lactate, (5) 2-2-hydroxybutyrate, (6) propionate, (7) formate, (8) butyrate, (9) 2-2- F hydroxyvalerate, (10) pyruvate, (11) isovalerate, (12) C102 CIO₂, (13) valerate, (14) Bro3_BrO (15) CH CI, (16) 2-2-oxovalerate, (17) NO2 NO₂ (18) ethylphosphonate, (19) trifluoroacetate, (20) azide, (21) Br NO (23) citramalate, (24) malate, (25) CO32- CO (30) a-ketoglutarate, a-ketoglutarate, wo²- Br (22) NO3-, (28) maleate, (29) SO42-,- fumarate, (33) oxaloacetate, (34) WO42, AsO SO² Mo0² (38) As 043- (44)1-I. 40 (26) malonate, (27) citraconitate, (31) C2042-C₂0² * (32) PO³ 1 (36) PO43- * (41) iso-citrate, (42) cis-aconitate, (43) trans-aconitate, > (37) phthalate,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Answer a The probable charge of pyruvate peak 1010 is 2 the probable charge of ...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Iodide ion is oxidized by hypochlorite ion in basic solution. I(aq) + ClO(aq) Cl(aq) + IO(aq) In 1.00 M NaOH at 25oC, the iodide-ion concentration (equal to the ClO concentration) at different times...
-
Consider the reaction of peroxydisulfate ion (S2O8-2) with iodide ion (I) in aqueous solution: S2O82- (aq) + 3 I (aq) - 2 SO42- (aq) + I3(aq) At a particular temperature the initial rate of...
-
The iodide ion catalyzes the decomposition of aqueous hydrogen peroxide, H2O2. This decomposition is believed to occur in two steps. H2O2 + I H2O + IO (elementary reaction) H2O2 + IO H2O + O2 + I...
-
Clara Hughes, who is pushing 5 0 , has medaled both in speedskating and road cycling ( and showing no signs of slowing down ) completed a training event where she biked 5 0 km east, stopped and rode...
-
For the conditions of Exercise 7 and the data given in Table 11.1, determine the value of R2, as defined by Eq. (11.5.26).
-
The McGregor Whisky Company is proposing to market diet scotch. The product will first be test-marketed for two years in southern California at an initial cost of $500,000. This test launch is not...
-
This problem helps you develop skill in recording transactions by using a companys actual account titles. Refer to the Amazon.com financial statements in Appendix A. Assume that Amazon.com completed...
-
The March 31, 2020, adjusted trial balance for Amusement Park Repair is shown below with accounts in alphabetical order. Required Preparation Component: a. Place an X in the space provided beside...
-
1. Explain what Donabedian Approach to standard says about : structure, process outcome 2. And how to Use the Donabedian Approach to standard: structure, process and outcometo explain the issues of...
-
Add a constraint to make sure the Rep_ID value entered in the BOOK_STORES table is a valid value contained in the STORE_REPS table. The Rep_ID columns of both tables were initially created as...
-
Isocratic separation with HPLC Teaching Assistant, an Excel spreadsheet that simulates a reversed-phase liquid chromatographic separation. Download the Excel file from SaplingPlus,...
-
Simulating a separation with a spreadsheet. Use the spreadsheet in Figure 25-39 to simulate the chromatograms for =0.75= 0.75 and =0.56= 0.56 in Figure 25-40. Figure 25-39 Figure 25-40 A 1...
-
Explain the third variable problem.
-
Dixon Company provides the following information about its product: Sales price per unit Variable cost per unit Total fixed costs Units sold What is the total contribution margin? $200 120 125,000...
-
Sunland Co.'s stockholders' equity at January 1, 2026 is as follows: Common stock, $10 par value; authorized 300,000 shares; Outstanding 227000 shares Paid-in capital in excess of par Retained...
-
The following information is provided by Smith Fabricating: Job 200 Job 201 Job 202 Direct Materials Cost Direct Labor Cost Labor Hours $500 $800 40 $350 $200 10 $1,000 $600 30 Smith Fabricating uses...
-
A rocket, initially at rest on the ground, accelerates straight upward from rest with constant acceleration m 53.9 The acceleration period lasts for time 7.00 s until the fuel is exhausted. After...
-
If there are no beginning retained earnings, net income of $ 3 0 , 3 0 0 , and ending retained earnings of $ 8 , 0 0 0 , how much are dividends?
-
Consider the permutations of 1, 2, 3, 4. The permutation 1432, for instance, is said to have one ascent - namely, 14 (since 1 < 4). This same permutation also has two descents - namely, 43 (since 4 >...
-
The executor of Gina Purcells estate has recorded the following information: Assets discovered at death (at fair value): Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Find the absolute and percent relative uncertainty and express each answer with a reasonable number of significant figures. (a) 9.23 ( 0.03) + 4.21 ( 0.02) - 3.26 ( 0.06) = ? (b) 91.3 ( 1.0) 40.3 (...
-
Verify the following calculations: (a) 3.1415( 0.0011) = 1.77243( 0.00031) (b) log[3.1415 ( 0.0011)] = 0.49714 ( 0.00015) (c) antilog[3.141.5 ( 0.0011)] = 1.3852( 0.0035) 103 (d) ln[3.1415 (...
-
Verify the following calculations: (a) 3.1415( 0.0011) = 1.77243( 0.00031) (b) log[3.1415 ( 0.0011)] = 0.49714 ( 0.00015) (c) antilog[3.141.5 ( 0.0011)] = 1.3852( 0.0035) 103 (d) ln[3.1415 (...
-
ABC company has accounts payable of 3 0 , 0 0 0 , long term debt of 2 5 , 0 0 0 , common stock with a value of 5 0 , 0 0 0 , sales in the amount of 1 0 0 , 0 0 0 , cash in the amount of 4 0 , 0 0 0...
-
Del Gato Clinic's cash account shows an $11,589 debit balance and its bank statement shows $10,555 on deposit at the close of business on June 30. a. Outstanding checks as of June 30 total $1,829. b....
-
Prepare a personal budget. If you are committed to a particular field, use the Internet (or other reliable information) to estimate your starting salary. Use the results of projects 2 and 3 to help...

Study smarter with the SolutionInn App


