Simulating a separation with a spreadsheet. Use the spreadsheet in Figure 25-39 to simulate the chromatograms for
Question:
Simulating a separation with a spreadsheet. Use the spreadsheet in Figure 25-39 to simulate the chromatograms for Ф=0.75Ф= 0.75 and Ф=0.56Ф= 0.56 in Figure 25-40.
Figure 25-39
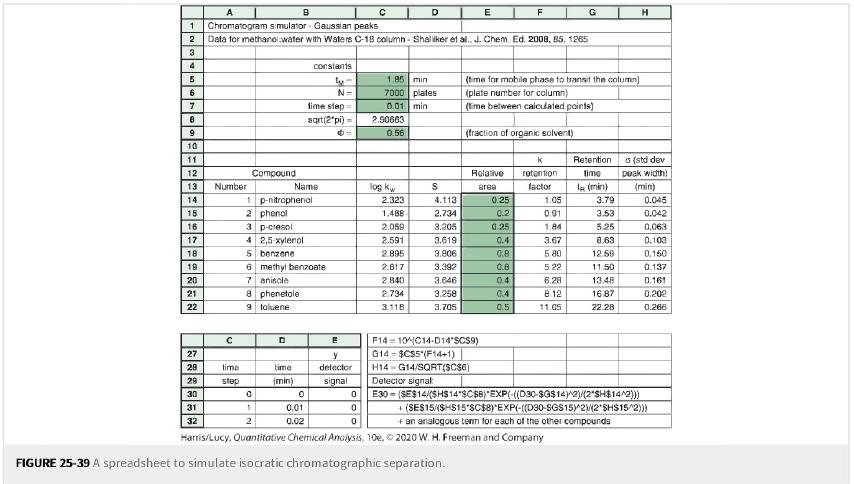
Figure 25-40

Transcribed Image Text:
A 1 Chromatogram simulator Gaussian peaks 2 Data for methanol water with Waters C-18 column-Shaliker et al., J. Chem. Ed. 2008, 85, 1265 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 328*** Number с time step Compound B 1 p-nitrophenol 2 phenol 8 phenetole 9 toluene 0 1 2 3 p-cresol 4 2,5-xylenol 5 benzene 6 methyl benzoate 7 aniscle D Name time (min) constants ly- N= time step= sqrt(2*pi)= 0 0.01 0.02 E y detector signal с 0 0 1.85 min 7000 plates 0.01 min 2.50663 0.56 log kw D 2.323 1.488 2.059 2.591 2.895 2.817 2,840 2.734 3.118 S 4.113 2.734 3.206 3.619 3,806 3.392 3.646 3,258 3.705 E 0 Hanis/Lucy, Quantitative Chemical Analysis, 10e, FIGURE 25-39 A spreadsheet to simulate isocratic chromatographic separation. F14104C14-D14"SCS9) G14 $CS5 (F14+1) H14 G14/SQRT($C$6) Detector signal (time for mobile phase to transit the column) (plate number for column) (time between calculated points) (fraction of organic solvent) Relative area 0.25 0.2 F 0.25 0.4 0.8 0.8 0.4 0.4 0.5 88 K retention: factor 1.05 0.91 1.84 G 3.67 5.80 5.22 6.28 8.12 11.05 Retention time 1 (min) 3.79 3.53 5.25 8.63 12.59 11.50 13.48 16.87 22.28 H (std dev peak width) (min) 0.045 0.042 0.063 0.103 0.150 0.137 0.161 0.202 0.266 E30-($E$14/(SH$14*$C$8) EXP(-((D30-$G$14)^2)/(2*SH$14^2))) +(SES15/(SHS15 SC$8)*EXP(-((D30-SGS15)^2(2 SHS15^2))) + an analogous term for each of the other compounds 2020 W. H. Freeman and Company
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
To simulate a separation with a spreadsheet you would need to create a table with the relevant param...View the full answer

Answered By

FELIX NYAMBWOGI
I have been tutoring for over 5 years, both in person and online. I have experience tutoring a wide range of subjects, including math, science, English, and history. I have also worked with students of all ages, from elementary school to high school.
In addition, I have received training in effective tutoring strategies and techniques, such as active listening, questioning, and feedback. I am also proficient in using online tutoring platforms, such as Zoom and Google Classroom, to effectively deliver virtual lessons.
Overall, my hands-on experience and proficiency as a tutor has allowed me to effectively support and guide students in achieving their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Use the counts in Figure 11.1 to complete Table 11.5 and answer the following questions. Table 11.1 Table 11.5 028 033 0.38 1/3 1/3 1/3 P(z) HIV No HIV Total +Test -Test Total 1,000,000
-
Use the flowchart in Figure to solve this problem. Assume that the master file and the transaction file are composed of the record numbers shown below in the sequence given. Record number 99 in each...
-
For the following exercises, use the graph in Figure 8, which shows the profit, y, in thousands of dollars, of a company in a given year, t, where t represents the number of years since 1980. Find...
-
Dwights preferences over beer and whiskey satisfy more is better, but are concave (thus violating the usual assumption of convexity). a. On a diagram, sketch what such indifference curves would look...
-
For the conditions of Exercise 7 and the data in Table 11.1, carry out a test of the following hypotheses: H0: 1 = 4, H1: 1 = 4.
-
What are operational efficiency and informational efficiency, and how do they differ in terms of trading markets?
-
Discuss the five key elements of the value management process.
-
Selected transactions for Niles Co. during March of the current year are listed in Problem 5-1B. In Problem 5-1B. The following selected transactions were completed by Niles Co. during March of the...
-
On March 1 2023, Blossom Winery Ltd. purchased a five-hectare commercial vineyard for $1,049,050. The total purchase price was based on appraised market values of the building, grapevines, and...
-
When a country has a comparative advantage in the production of a good, it means that it can produce this good at a lower opportunity cost than its trading partner. Then the country will specialize...
-
Consider the separation of inorganic and organic anions in Figure 26-7. a. What is the probable charge (Xn-) (X n- )of pyruvate (peak 1010), 2-2-oxovalerate (peak 1616), and maleate (peak 2828)? b....
-
Use Figure 25-33 for the following questions: a. What pHpH would be best for the separation of benzoic acid, 4-4-nitrophenol, and 3-3-methylbenzoic acid? b. What pHpH would be best for the separation...
-
Interest Rate Futures: Fair Value Hedge Fabric retailer Petren Corp. buys 300,000 yards of cloth each quarter at an average price of $3.00/yard. To finance these purchases, Petren sells $1,000 face...
-
On November 17, 1991, the Home News of central New Jersey used the bar chart in the accompanying figure to show the 19801991 passenger traffic trends for Newark International Airport. (a) Use these...
-
F is a partner in an investment partnership. She receives a payment equal to 10% of the partnerships income in exchange for advising services rendered to the partnership. In the current year, the...
-
You are given the following information about two stocks: (a) Calculate the mean and standard deviation for each stock. (b) Compare the mean, standard deviation, and coefficient of variation of each...
-
Your family started a new manufacturing business making outdoor benches for use in parks and outdoor venues two years ago. The business has been very successful, and sales are soaring. Because of...
-
Q is a 20 percent member in Lynn Properties, LLC. Because she is more involved than the other members in the LLC's activities, she receives a guaranteed payment from the LLC in the amount of $25,000...
-
What are the five special features of the Japanese system? Why haven't they been adopted in the United States?
-
Given the table below, about how much force does the rocket engine exert on the 4.0 kg payload? Distance traveled with rocket engine firing (m) Payload final velocity (m/s) 500 320 490 310 1020 450...
-
(a) Show that the formula mass of NaCl is 58.443 ( 0.0012) g/mol.
-
(a) Show that the formula mass of NaCl is 58.443 ( 0.0012) g/mol.
-
(a) Show that the formula mass of NaCl is 58.443 ( 0.0012) g/mol.
-
Image transcription text You encounter a split system that uses R-22 refrigerant and observe the following refrigeration parameters from the unit's control display. The unit is operating in cooling...
-
Calculating Present Values Imprudential, Inc., has an unfunded pension liability of $645 million that must be paid in 25 years. To assess the value of the firm's stock, financial analysts want to...
-
Describe the operating activities of each company noting similarities and difference between COCA COLA & PEPSICO. Identify two economy wide factors and industry wide factors that could impact on the...

Study smarter with the SolutionInn App


