Examine the procedure in Table 7-1 for the Fajans titration of Zn2+.Zn 2+ . Do you expect
Question:
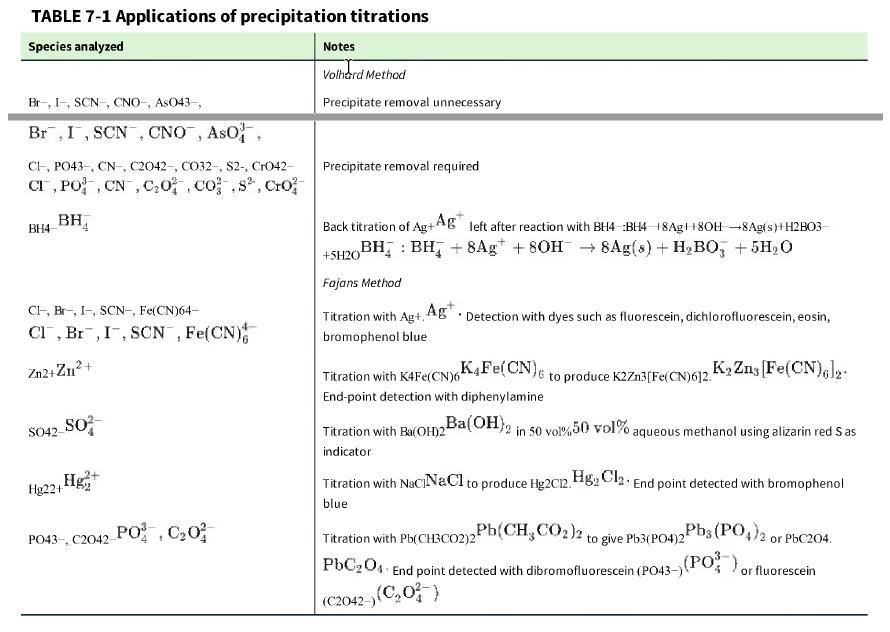
Examine the procedure in Table 7-1 for the Fajans titration of Zn2+.Zn2+. Do you expect the charge on the precipitate to be positive or negative after the equivalence point?
Table 7-1
Transcribed Image Text:
TABLE 7-1 Applications of precipitation titrations Species analyzed Br-, I-, SCN-, CNO-, AsO43-, Br, I, SCN, CNO, ASO, CI, PO43-, CN, C2042-, CO32-, S2-, CrO42- CI, PO, CN, C₂0, cos, Cro BH4 BH Cl, Br, I-, SCN-, Fe(CN)64- Cl, Br, I, SCN, Fe(CN) Zn2+Zn²+ SO42- SO² Hg₂+ Hg22+1 PO43-, C2042-PO, C₂0² Notes Volhard Method Precipitate removal unnecessary Precipitate removal required Back titration of Ag+Ag left after reaction with BH4 BH4 18Ag8OH-8Ag(s)+H2B03- BH BH₂ + 8Ag +80H 8Ag(s) + H₂BO3 + 5H₂O +5H201 Fajans Method Titration with Ag+, Ag. Detection with dyes such as fluorescein, dichlorofluorescein, eosin, bromophenol blue to produce K2Zn3[Fe(CN)612. K₂ Zns [Fe(CN)6]2. Ba(OH)2 in 50 vol%50 vol% aqueous methanol using alizarin red Sas K4Fe(CN)6 Titration with K4Fe(CN)6 End-point detection with diphenylamine Titration with Ba(OH)2 indicator Titration with NaciNaCl to produce Hg2012. HgCl₂ End point detected with bromophenol blue Pb(CH3CO2)2 to give Pb3(PO4)2 Titration with Pb(CH3CO2)2 PbC204 End point detected with dibromofluorescein (PO43-) (C2042-) (C₂0) Pb3(PO4)2 or PbC204. (PO), or fluorescein
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The Fajans titration of Zn2 produces a precipitate ...View the full answer

Answered By

Dennis Nyangau
I have been tutoring for several years now, and I absolutely love it! I love being able to help students one-on-one and see them succeed. It is so gratifying to see a student understand a concept that they were struggling with before. I also enjoy getting to know my students and helping them to reach their full potential.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Explain why f'(x) could be positive or negative at a point where f(x) > 0.
-
Explain why f(x) could be positive or negative at a point where f'(x) < 0.
-
In a rectangular coordinate system a positive point charge q = 6.00 X 10-9 C is placed at the point x = + 0.150 m, y = 0, and an identical point charge is placed at x = -0.150 m, y = 0. Find the x-...
-
According to a study by the American Pet Food Dealers Association, 63% of U.S. house-holds own pets. A report is being prepared for an editorial in the San Francisco Chronicle. As a part of the...
-
You have two possessions you would like to insure against theft or damage: your new bicycle, which cost you $800, and a painting you inherited, which has been appraised at $55,000. The painting is...
-
Ohno Industries purchased 12,000 of merchandise on February 1, 2022, subject to a trade discount of 10% and with credit terms of 3/15, n/60. It returned 3,000 (gross price before trade or cash...
-
When using statistical sampling for attributes, an auditor should assess control risk at the maximum assuming: a. The sample rate of deviation plus the allowance for sampling risk is less than the...
-
Superior Uniforms produces uniforms. The company allocates manufacturing overhead based on the machine hours each job uses. Superior Uniforms reports the following cost data for the past year:...
-
If a river is discharging at 9,000 cubic feet per second at itsdelta, calculate the rivers velocity if the width is 40 meters andthe depth is 3.5 meters. Please express your answer usingEnglish units...
-
You purchased Enron stock at a price of $30 per share. Its price was $20 after six months and the company declared bankruptcy at the end of the next six months. The realized return over the last year...
-
What is wrong with this procedure? According to Table 7-1, carbonate can be measured by a Volhard titration. Removal of the precipitate is required. To analyze an unknown solution of Na 2 CO 3 ,Na2CO...
-
Write the Kb K b reaction of CN-CN - Given that the Ka K a , value for HCN HCN is 6.2x10 -10 , 6.2x10 -10 calculate Kb K b for CN-CN - .
-
Presented below is information related to P. J. Harvey Company for 2008. Retained earnings balance, January 1, 2008.................................................$980,000 Sales for the...
-
18. Which of the following diseases is transmitted via aerosols? a. Psittacosis b. Shigellosis c. Leptospirosis d. Giardiasis 19. Which of the following organisms is used in the production of cheese?...
-
1. The atomic number equals the number of a. protons. b. neutrons. c. electrons. d. protons and neutrons. 2. The atomic weight is equal to the sum of a. neutrons. b. protons and neutrons. c. neutrons...
-
4. Protons are a. located in the shells. b. part of the atomic nucleus. c. negatively charged. d. uncharged particles 5. Particles of an atom located in the outermost shell and available for chemical...
-
General Ledger Accounts; Trial Balance Diane Bernick opened Bernick's Consulting. Transactions and related journal entries for January are shown. Jan. 1 Bernick invested cash in the business,...
-
7. A chemical bond in which electrons are equally shared is a(n) a. ionic bond. b. polar covalent bond. c. nonpolar covalent bond. d. hydrogen bond. 8. The transfer of electrons in a chemical bond...
-
An example of how the APT language would be used on a part is shown in Figure 32-35. Essentially, it is necessary to tell the computer the location of a center of the bolt hole circle or one of the...
-
-4 1 9. Let A = Find A-1, (A") and verify that (A")= (A-1)".
-
What is the difference between a false positive and a false drop? If it is essential that no relevant information be missed by an information retrieval query, is it acceptable to have either false...
-
What is the difference between e-commerce and m-commerce?
-
The head of institutional research at a university believed that the mean age of full-time students was declining. In 1995, the mean age of a full-time student was known to be 27.4 years. After...
-
Flounder Company reported the following information for 2019: October November December Budgeted sales $300,000 $334,000 $378,000 Budgeted purchases $120,000 $127,000 $144,500 All sales are on...
-
Concord Co. decides at the beginning of 2025 to adopt the FIFO method of inventory valuation. Concord had used the LIFO method for financial reporting since its inception on January 1, 2023, and had...
-
An experimental rocket sled accelerates from 12.0 m/s to 280 m/s in 4.70 seconds. How far did it travel in this time?

Study smarter with the SolutionInn App


