What is wrong with this procedure? According to Table 7-1, carbonate can be measured by a Volhard
Question:
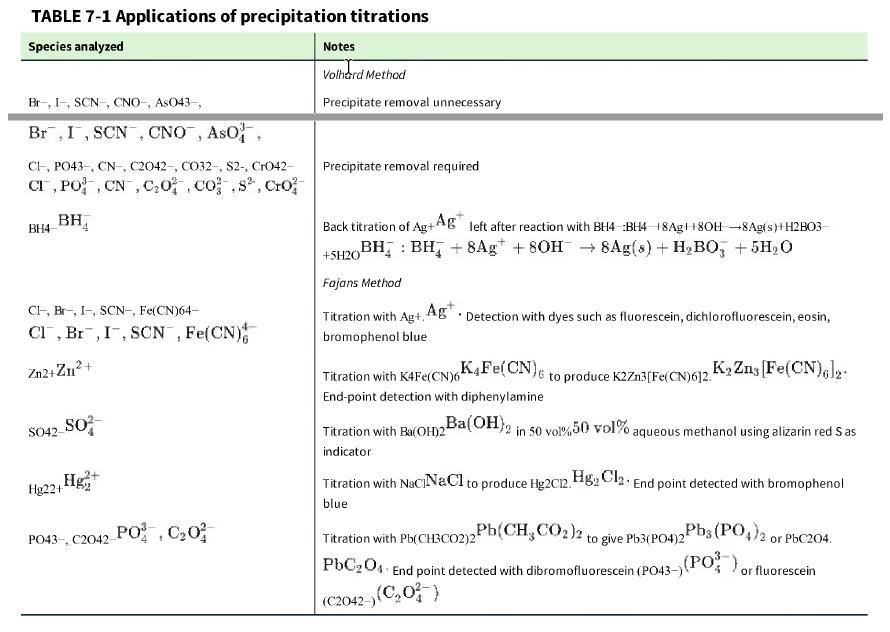
What is wrong with this procedure? According to Table 7-1, carbonate can be measured by a Volhard titration. Removal of the precipitate is required. To analyze an unknown solution of Na2CO3,Na2CO3, I acidified the solution with freshly boiled and cooled HNO3 HNO3 to give ~0.5 M HNO3. ~0.5 M HNO3 Then I added excess standard AgNO3, AgNO3 but no Ag2CO3 Ag2CO3 precipitate formed. What happened?
Table 7-1
Transcribed Image Text:
TABLE 7-1 Applications of precipitation titrations Species analyzed Br-, I-, SCN-, CNO-, AsO43-, Br, I, SCN, CNO, ASO, CI, PO43-, CN, C2042-, CO32-, S2-, CrO42- CI, PO, CN, C₂0, cos, Cro BH4 BH Cl, Br, I-, SCN-, Fe(CN)64- Cl, Br, I, SCN, Fe(CN) Zn2+Zn²+ SO42- SO² Hg₂+ Hg22+1 PO43-, C2042-PO, C₂0² Notes Volhard Method Precipitate removal unnecessary Precipitate removal required Back titration of Ag+Ag left after reaction with BH4 BH4 18Ag8OH-8Ag(s)+H2B03- BH BH₂ + 8Ag +80H 8Ag(s) + H₂BO3 + 5H₂O +5H201 Fajans Method Titration with Ag+, Ag. Detection with dyes such as fluorescein, dichlorofluorescein, eosin, bromophenol blue to produce K2Zn3[Fe(CN)612. K₂ Zns [Fe(CN)6]2. Ba(OH)2 in 50 vol%50 vol% aqueous methanol using alizarin red Sas K4Fe(CN)6 Titration with K4Fe(CN)6 End-point detection with diphenylamine Titration with Ba(OH)2 indicator Titration with NaciNaCl to produce Hg2012. HgCl₂ End point detected with bromophenol blue Pb(CH3CO2)2 to give Pb3(PO4)2 Titration with Pb(CH3CO2)2 PbC204 End point detected with dibromofluorescein (PO43-) (C2042-) (C₂0) Pb3(PO4)2 or PbC204. (PO), or fluorescein
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Thorough explanation The procedure is incorrect because the carbona...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
What is wrong with this "proof" that all horses are the same color? Let P(n) be the proposition that all the horses in a set of n horses are the same color. Inductive Step: Assume that P(k) is true,...
-
What is wrong with this "proof"? "Theorem" For every positive integer n, if x and y are positive integers with max(x, y) = n, then x = y. Basis Step: Suppose that n = 1. If max(x, y) = 1 and x and y...
-
What is wrong with this statement? ( )-inx sima cos (tan x) cos cos
-
Suppose, in the New Monetarist model, that there is deficient financial liquidity. If the fiscal authority were to engineer a tax cut, financed by an increase in the quantity of government debt, with...
-
Say whether each of the following scenarios describes an insurance problem caused by adverse selection or by moral hazard. a. People who have homeowners insurance are less likely than others to...
-
A published study of a chemical reaction A ? P, indicates that if the reactor initially contains A at a concentration C A0 (g/L) and the reaction temperature, T, is kept constant, then the...
-
A confidence interval for 1 is to be constructed from a sample of 20 points. How many degrees of freedom are there for the critical value?
-
Variable price and usage variances and fixed manufacturing overhead cost variances Kemp Tables, Inc. (KTI), makes picnic tables of 2 3 4 planks of treated pine. It sells the tables to large retail...
-
Williams Products Inc. manufactures and sells a number of items, including school knapsacks. The company has been experiencing losses on the knapsacks for some time, as shown by the contribution...
-
Assume you've generated the following information about the stock of Bufford's Burger Barns: The company's latest dividends of $4 a share are expected to grow to $4.32 next year, to $4.67 the year...
-
Maximize or minimize the given objective function f subject to the given constraints. Maximize f = 30x 1 + 10x 2 in the region in Prob. 5. Data from Prob. 5 Describe and graph the regions in the...
-
Examine the procedure in Table 7-1 for the Fajans titration of Zn2+.Zn 2+ . Do you expect the charge on the precipitate to be positive or negative after the equivalence point? Table 7-1 TABLE 7-1...
-
Refer to Exercise 15.16. a. Describe how the varieties of plants and planting densities would be randomly assigned to the plots of land. b. Compute the relative efficiency of the randomized block...
-
Explain how the income of each type of business entity is taxed, specifying whether the entity, the owner, or both, are taxed. Analyze the advantages and disadvantages of each entity in this context....
-
Cullumber Marine Corporation s unadjusted trial balance includes the following balances ( assume normal balances ) : Accounts receivable $ 1 8 4 0 0 0 0 Allowance for doubtful accounts $ 3 3 7 0 0...
-
In C Program,electronics, voltage drop is the decrease of electric potential along the path of a current flowingin a circuit. Voltage drops in the internal resistance of the source, across...
-
Consider a monoatomic gas at STP whose N particles are confined to move only over a 2D square (rather than a 3D cube). The spectrum of these particles are given by = 2ma ;(n /2 + n) where m is the...
-
3) Equity Investment transactions (4 marks) Riley Holdings, a public company, purchased the following investments on September 1, 2024: 8,000 common shares of Jasper Public Co., which trade on the...
-
Let S = {(x, y, z): x2 / (2 + y2 / b2 + z2 / c2 ( 1}. Evaluate (xy + xz + yz) dV.
-
Q:1 Take any product or service offered in Pakistan and apply all determinents of customer Perceived value ?
-
A specimen of medium-carbon steel having an initial diameter of 0.503 in was tested in tension using a gauge length of 2 in. The following data were obtained for the elastic and plastic states: Note...
-
A firework charge is detonated many meters above the ground. At a distance of 400 m from the explosion, the acoustic pressure reaches a maximum of 10.0 N/m2. Assume that the speed of sound is...
-
One kilo mole Ar and one kilo mole O2 is heated up at a constant pressure of 100 kPa to 3200 K, where it comes to equilibrium. Find the final mole fractions for Ar, O2, and O.
-
time complexity of the following algorithm forn-1 to n-1 do for je +1 to n do Print & for Kn-3 to n+4 do print k
-
Quentin is considering purchasing an investment. He expects that the investment will pay him $2,600 at the end of every year for 9 years. In addition, he expects to receive $34,752 at the end of the...
-
Oriole Company receives $339,600 when it issues a $339,600, 10% mortgage note payable to finance the construction of a building at December 31, 2022. The terms provide for annual installment payments...

Study smarter with the SolutionInn App


