Use Figure 25-33 for the following questions: a. What pHpH would be best for the separation of
Question:
Use Figure 25-33 for the following questions:
a. What pHpH would be best for the separation of benzoic acid, 4-4-nitrophenol, and 3-3-methylbenzoic acid?
b. What pHpH would be best for the separation of benzoic acid, 3-3-methylbenzoic acid, and 4-4-methylaniline?
c. What pHpH would be best for separation of 4-4-nitrophenol, 4-4-methylaniline, and codeine on a typical C18-silica C18-silica column?
Figure 25-33
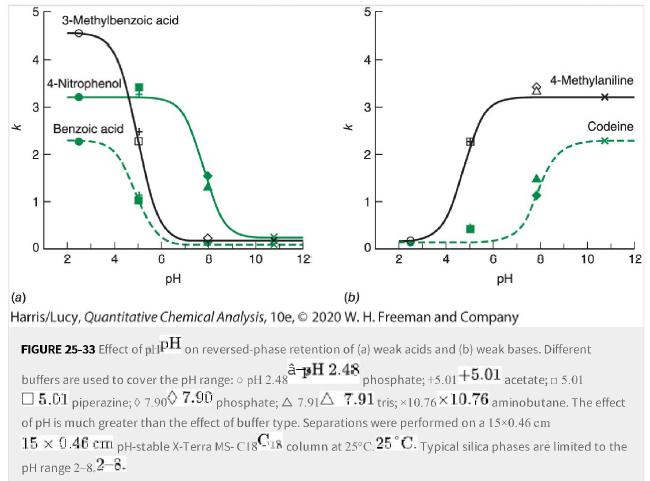
Transcribed Image Text:
K 3 2 1 3-Methylbenzoic acid 4-Nitrophenol Benzoic acid 2 G 0 10 12 pH K 5 4 3 2 1 0 I 2 4-Methylaniline Codeine 10 12 pH (b) (a) Harris/Lucy, Quantitative Chemical Analysis, 10e, 2020 W. H. Freeman and Company FIGURE 25-33 Effect of plPH on reversed-phase retention of (a) weak acids and (b) weak bases. Different à pH 2.48 buffers are used to cover the pH range: o pH 2.48 phosphate; +5.01+5.01 acetate; n 5.01 5.01 piperazine; 7.900 7.90 phosphate; A 7.91A 7.91 tris; 10.76×10.76 aminobutane. The effect of pH is much greater than the effect of buffer type. Separations were performed on a 15x0.46 cm 15 x 0.46 cm pH-stable X-Terra MS-C1818 column at 25°C 25 °C. Typical silica phases are limited to the pH range 2-8.2-8.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Answer a The best pH for the separation of benzoic acid 44nitroph...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Refer to below graph Figure 141 to answer the following questions a What is the Federal Fund Rate at the initial level of reserves supply b How does the Fed hit its federal funds rate target of 2
-
Select all the correct option for the following questions 1. Money a. Is more efficient than barter. b. Makes trades easier. c. Allows greater specialization. d. All of the above are correct. 2....
-
Highlight the correct option for the following questions 31. When a firm expands in the production process to supply its own raw materials and intermediate products or to provide its own sales or...
-
Hybrid Corporation began operations in 2018 and during that year purchased equity investments. It owns less than 20% of the voting shares for all companies whose stock it purchased. The year-end cost...
-
Show that the M.L.E. of 2 in the general linear model is given by Eq. (11.5.7).
-
Photographic laboratories recover and recycle the silver used in photographic film. Stikine River Photo is considering purchase of improved equipment for their laboratory at Telegraph Creek. Here is...
-
Peppertree Copy Center had accounts receivable of \(\$ 20,000\) at the beginning of the year and \(\$ 50,000\) at year-end. Revenue for the year totaled \(\$ 110,000\). How much cash did Peppertree...
-
The following data were taken from the balance sheet of Beatty Company: a. Determine for each year (1) the working capital, (2) the current ratio, and (3) the quick ratio. Round ratios to one decimal...
-
Define supply chain management and what it involves. Explain each of the 10 key elements of supply chain management and the importance of why each should be effectively managed.
-
Broker Sonya Wong is currently trying to maximize her profit in the bond market. Four bonds are available for purchase and sale at the bid and ask prices shown in the file P04_97.xlsx. Sonya can buy...
-
Simulating a separation with a spreadsheet. Use the spreadsheet in Figure 25-39 to simulate the chromatograms for =0.75= 0.75 and =0.56= 0.56 in Figure 25-40. Figure 25-39 Figure 25-40 A 1...
-
The figure shows reversed-phase retention data for three compounds. a. Identify whether compounds A, B, and C are weak acids or bases. For each compound, what is the pKap K a and the retention factor...
-
If P (A) = 0.75, P (B) = 0.4, and P (A and B) = 0.25, find P (A or B).
-
1. What management approach does Riverside State Hospital's style of man- agement most resemble? 2. How does the management approach influence communication at the hospital? 3. Should Ellis be held...
-
G & L Plastic Molders paid consulting fee of $73,000 last week for feedback on a new project. This week the company is trying to decide whether the new project should be undertaken. Should the...
-
Langton, N., Robbins, S. P., & Judge, T. A. (2019). Organizational behaviour: Concepts, controversies, applications (8th Canadian ed.). Pearson Canada Inc. Chapter 11: Leadership Chapter 12: Decision...
-
10x = solve (100)(1011) (109)
-
The electric field 0 . 295 m from a very long uniform line of charge is 8 30 N / C Part A How much charge is contained in a section of the line of length 2 . 70 cm ? Express your answer in coulombs.
-
Give a recursive definition for the set of all (a) Positive even integers (b) Nonnegative even integers
-
Inexhaustible collections of ONPOs are not required to be capitalized or depreciated, if certain criteria are met. Why is this so, and what accounting and reporting recognition, if any, is required...
-
What is the true mass of water weighed at 24C in the air if the apparent mass is 1.0346 0.0002 g? The density of air is 0.0012 0.0001g/mL and the density of balance weights is 8.0 0.5 g/mL. The...
-
Round each number as indicated: (a) 1.236 7 to 4 significant figures (b) 1.238 4 to 4 significant figures (c) 0.135 2 to 3 significant figures (d) 2.051 to 2 significant figures (e) 2.005 0 to 3...
-
Twelve dietary iron tablets were analyzed by the gravimetric procedure in Section 1-4 and the final mass of Fe2O3 (FM 159.688) was 0.2774 0.0018 g. Find the average mass of Fe per tablet.(Relative...
-
q11. XYZ Corporation issued 12% bonds, dated January 1, with a face amount of $470 million on January 1, 2024, for $420,208,482. The bonds mature on December 31, 2033 (10 years). For bonds of similar...
-
Labeau Products, Limited, of Perth, Australia, has $25,000 to invest. The company is trying to decide between two alternative uses for the funds as follows: Investment required Annual cash inflows...
-
Use the 2015 tax table to find the income tax for a taxpayer with taxable income of $81,083 filing as head of household. Use the 2015 tax table to find the income tax for a taxpayer with taxable...

Study smarter with the SolutionInn App


