In x-ray studies of crystalline peptides. Linus Pauling and Robert Corey found that the CON bond in
Question:
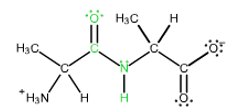
In x-ray studies of crystalline peptides. Linus Pauling and Robert Corey found that the CON bond in the peptide link is intermediate in length (1.32 A) between a typical C?N single bond (1.491) and a C=N double bond (1.27 A). They also found that the peptide bond is planar (all four atoms attached to the CN group are located in the same plane) and that the two a-carbon atoms attached to the CON are always trans to each other (on opposite sides of the peptide bond).
The shortened length of the C?N bond can be explained by the existence of an alternative resonance structure for the peptide bond. Modify the structure of the dipeptide Ala-Ala, shown at the right, to demonstrate the other resonance structure that exists. (Modify only the peptide bond. Be sure to add formal charges if needed. If you mistakenly modify any other part of the structure you can recover the original structure by clicking on the double red arrow (0) at the bottom of the drawing module.
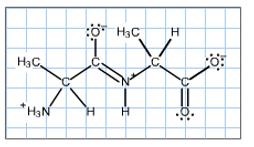
The double bond character added to the C?N bond by resonance results in restricted rotation about the C?N bond, explaining why the atoms about the peptide bond all lie in a plane. In the structure at the right, select all of the atoms and bonds that lie in the peptide plane by clicking on them. (When selected, and atom or bond will turn green. If one is selected by mistake, click on it again to deselect.)