0.5pts) 1. At ice temperatures (below 5 C), the vapor pressure of water is negligible. The...
Fantastic news! We've Found the answer you've been seeking!
Question:

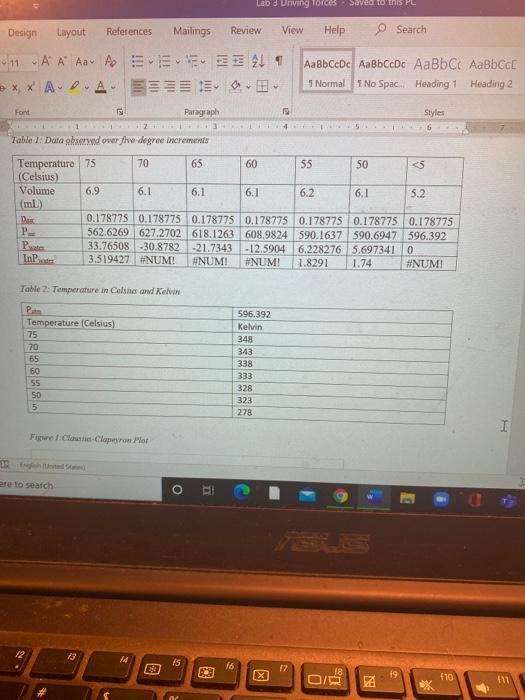
Transcribed Image Text:
0.5pts) 1. At ice temperatures (below 5 "C), the vapor pressure of water is negligible. The volume at that point is due to the air alone, within the limits of the measurements. Using the gas law equation, the number of moles of air in the cylinder is calculated: (Use R = 62.4 L*torr*Kmol") %3D PVeold RTeld 13285.0141 Using the corrected value for volume, the lowest measured temperature and the atmospheric pressure, calculate the number of moles of trapped air. Lab 3 Driving forces Saved to this PC Design Layout References Mailings Review View Help O Search 市、而。同 1 圓圳面 。,田。 11 A A Aav Ap AaBbCcDc AaBbCcDc AaBbC AaBbCE 1 No Spac. Heading 1 e x, x A- A. 1 Normal Heading 2 Font Paragraph Styles 4 Table 1: Data observed over five-degree increments Temperature 75 (Celsius) Volume (mL) 70 65 60 55 50 <5 6.9 6.1 6.1 6.1 6.2 6.1 5.2 0.178775 0.178775 0.178775 0.178775 0.178775 0.178775 0.178775 562.6269 627.2702 618.1263 608.9824 590.1637 590.6947 | 596.392 33.76508 -30.8782 -21.7343 -12.5904 6.228276 5.697341 0 3.519427 #NUM! Bais P Pta InP #NUM! #NUM! 1.8291 1.74 #NUM! Table 2: Temperature in Celsna and Kelvin P 596.392 Temperature (Celsius) Kelvin 75 348 70 343 65 338 60 333 55 328 50 323 278 Figre Claustus-Clapeyron Plot d St ere to search TSLES 13 14 f6 E3 18 19 f10 411 图 0.5pts) 1. At ice temperatures (below 5 "C), the vapor pressure of water is negligible. The volume at that point is due to the air alone, within the limits of the measurements. Using the gas law equation, the number of moles of air in the cylinder is calculated: (Use R = 62.4 L*torr*Kmol") %3D PVeold RTeld 13285.0141 Using the corrected value for volume, the lowest measured temperature and the atmospheric pressure, calculate the number of moles of trapped air. Lab 3 Driving forces Saved to this PC Design Layout References Mailings Review View Help O Search 市、而。同 1 圓圳面 。,田。 11 A A Aav Ap AaBbCcDc AaBbCcDc AaBbC AaBbCE 1 No Spac. Heading 1 e x, x A- A. 1 Normal Heading 2 Font Paragraph Styles 4 Table 1: Data observed over five-degree increments Temperature 75 (Celsius) Volume (mL) 70 65 60 55 50 <5 6.9 6.1 6.1 6.1 6.2 6.1 5.2 0.178775 0.178775 0.178775 0.178775 0.178775 0.178775 0.178775 562.6269 627.2702 618.1263 608.9824 590.1637 590.6947 | 596.392 33.76508 -30.8782 -21.7343 -12.5904 6.228276 5.697341 0 3.519427 #NUM! Bais P Pta InP #NUM! #NUM! 1.8291 1.74 #NUM! Table 2: Temperature in Celsna and Kelvin P 596.392 Temperature (Celsius) Kelvin 75 348 70 343 65 338 60 333 55 328 50 323 278 Figre Claustus-Clapeyron Plot d St ere to search TSLES 13 14 f6 E3 18 19 f10 411 图
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The following data give the vapor pressure of water at various temperatures. Transform the data, using ln (P) for the dependent variable and 1/T for the independent variable. Carry out the linear...
-
The vapor pressure of water is 17.5 mmHg at 20.0C and 355.1 mmHg at 80.0C. Calculate the boiling point of water at 760.0 mmHg.
-
The vapor pressure of a volatile liquid can be determined by slowly bubbling a known volume of gas through the liquid at a given temperature and pressure. In an experiment, a 5.40-L sample of...
-
Write a program RecoverSignal that will read the binary file written by StoreSignal, as described in the previous exercise. Display the integer values that the data represents on the screen.
-
Assume that you have a sample of n1 = 8, with the sample mean 1 = 42, and a sample standard deviation S1 = 4, and you have an independent sample of n2 = 15 from another population with a sample mean...
-
Graph using addition of ordinates. Then check your work using a graphing calculator. y = 2 sin x + cos 2x
-
The Wide World of Fluids article titled "A Sailing Ship without Sails,". Determine the magnitude of the total force developed by the two rotating cylinders on the Flettner "rotor-ship" due to the...
-
On January 6, Petro Co. sells merchandise on account to Chose Inc. for $9,200, terms 1/10, n/30. On January 16, Chose pays the amount due. Instructions Prepare the entries on Petro Co.s books to...
-
Asking an applicant/candidate if they have children, or plan on having children, is an illegal question. Does anyone know how organizations ascertain information like this without asking the illegal...
-
6. You have implemented a butterfly spread whereby the strategy will make money if the stock ends up between $44 and $56 (8 months from now). The stock currently trades at $48, its historical...
-
When 10 British Army soldiers on a 10-day training exercise descended into Low's Gully, a narrow chasm that cuts through Mt. Kinabalu in Borneo, each knew "the golden rule for such expeditions -...
-
Saying that a quantitative trait exhibits a continuum means that a. the numerical value for the trait increases with the age of the individual. b. environmental effects are additive. c. the...
-
The government has today imposed a price ceiling on gasoline purchased at the pump. As one member of Congress said, It was done to keep gasoline prices within the reach of the poor and the...
-
An antibiotic is a drug that kills or inhibits the growth of microorganisms. The use of antibiotics has been of great importance in the battle against many infectious diseases caused by...
-
With regard to the CRISPR-Cas system that defends bacteria against bacteriophage attack, what happens during the adaptation, expression, and interference phases? When a bacterium is exposed to a...
-
A hypothetical base sequence of an RNA molecule is 5AUUUGCCCUAGCAAACGUAGCAAACG3 Using two of the three underlined parts of this sequence, draw two possible stem-loop structures that might form in...
-
Find the coordinates of the vertex for the para function. f(x)=-7(x-3)2 5
-
Use Stokes' Theorem to evaluate f(y+sin x) dx+(z+cos y) dy+rdz, where C is the rve r(t) = (sint, cost, sin 2t), t = [0, 2].
-
A 1.50-g sample of nitrous oxide (an anesthetic, sometimes called laughing gas) contains 2.05 1022 N2O molecules. How many nitrogen atoms are in this sample? How many nitrogen atoms are in 1.00 g of...
-
Chlorine can be prepared by oxidizing chloride ion (from hydrochloric acid) with potassium dichromate, K2Cr2O7, which is reduced to Cr3+. Write the balanced equation for the reaction.
-
Predict the order of increasing vapor pressure at a given temperature for the following compounds: a. CH3CH2CH2CH2OH b. CH3CH2OCH2CH3 c. HOCH2CH2CH2OH Explain why you chose this order.
-
In a perfectly contestable market, inefficiency does not exist. Explain why.
-
A perfectly competitive market is by definition perfectly contestable, but a perfectly contestable market is not necessarily a perfectly competitive market. Comment on this statement.
-
Why is it important to be clear about how costs can be classified to consider the degree of contestability of a market?
Financial Resource Management For Nonprofit Organizations 1st Edition - ISBN: 0133162990 - Free Book

Study smarter with the SolutionInn App



