1. A cycle consisting of one constant pressure, one constant volume and two isentropic processes is...
Fantastic news! We've Found the answer you've been seeking!
Question:
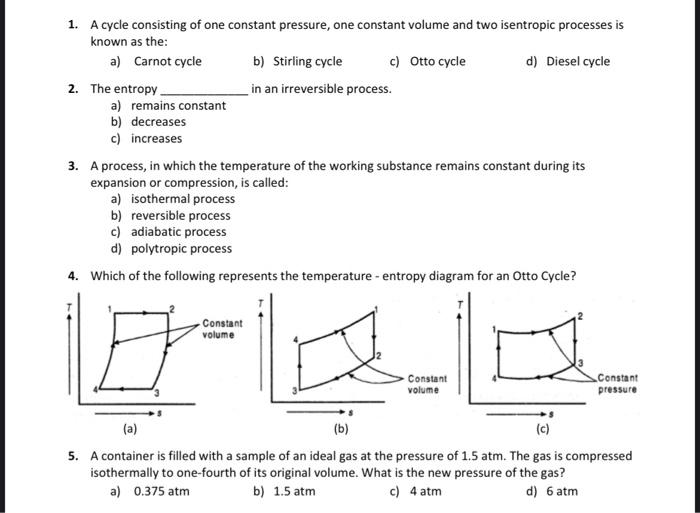
Transcribed Image Text:
1. A cycle consisting of one constant pressure, one constant volume and two isentropic processes is known as the: a) Carnot cycle 2. The entropy. a) remains constant b) decreases c) increases b) Stirling cycle in an irreversible process. c) Otto cycle Constant volume 3. A process, in which the temperature of the working substance remains constant during its expansion or compression, is called: a) isothermal process b) reversible process c) adiabatic process d) polytropic process 4. Which of the following represents the temperature - entropy diagram for an Otto Cycle? d) Diesel cycle Constant volume Constant pressure (a) (b) (c) 5. A container is filled with a sample of an ideal gas at the pressure of 1.5 atm. The gas is compressed isothermally to one-fourth of its original volume. What is the new pressure of the gas? a) 0.375 atm b) 1.5 atm c) 4 atm d) 6 atm 1. A cycle consisting of one constant pressure, one constant volume and two isentropic processes is known as the: a) Carnot cycle 2. The entropy. a) remains constant b) decreases c) increases b) Stirling cycle in an irreversible process. c) Otto cycle Constant volume 3. A process, in which the temperature of the working substance remains constant during its expansion or compression, is called: a) isothermal process b) reversible process c) adiabatic process d) polytropic process 4. Which of the following represents the temperature - entropy diagram for an Otto Cycle? d) Diesel cycle Constant volume Constant pressure (a) (b) (c) 5. A container is filled with a sample of an ideal gas at the pressure of 1.5 atm. The gas is compressed isothermally to one-fourth of its original volume. What is the new pressure of the gas? a) 0.375 atm b) 1.5 atm c) 4 atm d) 6 atm
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these mechanical engineering questions
-
An ideal gas goes through a cycle consisting of isothermal, polytropic, and adiabatic lines, with the isothermal process proceeding at the maximum temperature of the whole cycle. Find the efficiency...
-
An ideal gas goes through a cycle consisting of alternate isothermal and adiabatic curves (Fig. 2.2). The isothermal processes proceed at the temperatures T1, T2, and T3. Find the efficiency of such...
-
An ideal gas goes through a cycle consisting of (a) Isochoric, adiabatic, and isothermal lines; (b) Isobaric, adiabatic, and isothermal lines, with the isothermal process proceeding at V the minimum...
-
Evaluate each expression if possible. (-3)4
-
St. Genevieve Petroleum Company is an independent oil producer in Baton Parish, Louisiana. In February, company geologists discovered a pool of oil that tripled the companys proven reserves. Prior to...
-
What problem can occur when a database is processed by more than one user?
-
Who can sign a pleading?
-
On August 3, 2013, the date of incorporation, Quinn Company accepts separate subscriptions for 1,000 shares of $100 par preferred stock at $104 per share and 9,000 shares of no-par, no-stated-value...
-
1. What is crisis communication? Please explain the role of public relations in crisis communication. 2. Please examine Exxon company's crisis management regarding the Exxon Valdez oil spill. 3. What...
-
Consider the operations of a fast-food restaurant serving pre-made pasta dishes to its guests. The operation consists of four steps. Each customer is first greeted and offered a menu and a tray,...
-
The Addams family has an annual earned income of $78,700, which is around the median US household income. If the Addams family has long-term capital gains of $50,350 in 2023, how much capital gains...
-
When it comes to security what types of risk are associated with positive outcomes and what industries would you expect this type of risks? On the other side what role do you think positive risk...
-
Question 2 Your superior at the Money Management Company you work for has just analyzed your portfolio. He instructs you to reduce cash holdings and to reduce the overall portfolio beta. In order to...
-
7. Consider the following time series OLS regression t 3.41 +2.72x1t - 5.33x2t = DW 1.60 and n = 60 where t indexes the period of observation; DW denotes the Durbin-Watson statistic; and n is the...
-
DATA: After making sure that the cash-flow models for all scenarios are correct according to the solution provided for Homework-2, use the NCF After Tax (Column # 14) with an i* of 8% per year to;...
-
what ways do implicit biases and cognitive heuristics intersect with the multifaceted layers of organizational culture, and how can interventions leveraging principles from behavioral economics and...
-
Consider the following observation: The stopping distance for a car is much shorter than for a train going the same speed?. For the following questions, assume purely horizontal motion with the...
-
The Dow Jones Industrial Average reached a high of $ 7801.63 on December 29, 1997. Recall from Example 18.4 that it reached a high of $ 1003 on November 14, 1972. The Consumer Price Index for...
-
(a) What does the term paramagnetic mean? (b) How can one determine experimentally whether a substance is paramagnetic? (c) Which of the following ions would you expect to be paramagnetic: O2+, N22-,...
-
Write the chemical formula for each of the following compounds, and indicate the oxidation state of the group 6A element in each: (a) Sulfur tetrachloride (b) Selenium trioxide (c) Sodium thiosulfate...
-
Predict the type of radioactive decay process for the following radionuclides: (c) Phosphorus-32, (d) chlorine-39. B,
-
The rotor shown in Fig. 9.44 (a) is balanced temporarily in a balancing machine by adding the masses \(m_{1}=m_{2}=90 \mathrm{~g}\) in the plane \(A\) and \(m_{3}=m_{4}=90 \mathrm{~g}\) in the plane...
-
A turbine rotor is run at the natural frequency of the system. A stroboscope indicates that the maximum displacement of the rotor occurs at an angle \(229^{\circ}\) in the direction of rotation. At...
-
The cylinders of a four-cylinder in-line engine are placed at intervals of \(300 \mathrm{~mm}\) in the axial direction. The cranks have the same length, \(100 \mathrm{~mm}\), and their angular...

Study smarter with the SolutionInn App


