1. Complete the extraction scheme for the separation of 1,2-dichlorobenzene, phenol (pKa = 10), and pyridine....
Fantastic news! We've Found the answer you've been seeking!
Question:
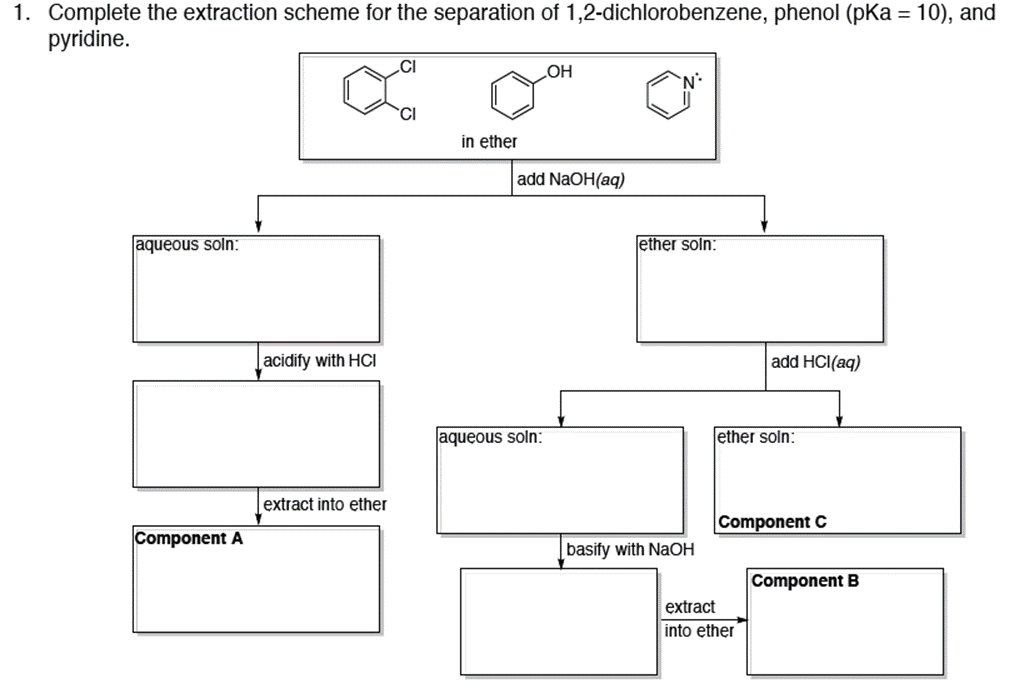
Transcribed Image Text:
1. Complete the extraction scheme for the separation of 1,2-dichlorobenzene, phenol (pKa = 10), and pyridine. .CI OH in ether add NaOH(aq) aqueous soln: ether soln: |acidify with HCI add HCI(aq) aqueous soln: ether soln: extract into ether Component C Component A basify with NaOH Component B extract into ether 1. Complete the extraction scheme for the separation of 1,2-dichlorobenzene, phenol (pKa = 10), and pyridine. .CI OH in ether add NaOH(aq) aqueous soln: ether soln: |acidify with HCI add HCI(aq) aqueous soln: ether soln: extract into ether Component C Component A basify with NaOH Component B extract into ether
Expert Answer:
Related Book For 

Fundamentals of Analytical Chemistry
ISBN: 978-0495558286
9th edition
Authors: Douglas A. Skoog,Donald M. West, F. James Holler, Stanley R. Crouch
Posted Date:
Students also viewed these accounting questions
-
The molar absorptivity for aqueous solutions of phenol at 211 nm is 6.17 ( 103 L cm-1 mol-1. Calculate the permissible range of phenol concentrations if the transmittance is to be less than 85% and...
-
The logarithm of the molar absorptivity of phenol in aqueous solution is 3.812 at 211 nm. Calculate the range of phenol concentrations that can be used if the absorbance is to be greater than 0.150...
-
A mass of 400(lbm) of 35-wl-% aqueous NaOH solution at 130(F) is mixed with 175(lbm) of 10-wt-% solution at 200(F). (a) What is the heat effect if the final temperature is 80(F)? (b) If the mixing is...
-
As the Developing market of Southern Africa there is endless growth opportunities and constant ask for high growth pushed by the business, this growth comes normally in a very erratic way but as...
-
You have decided that you will sell off your house, which is currently valued at $300,000 when it appreciates in value to $450,000. If houses are appreciating at an average annual rate of 4.5% in...
-
Sally incurred a 90-mile round-trip commute every day, mainly because she could not get along with her super-visor at the sales office located fourmiles fromher home. Sally works under a one-year...
-
The comparative financial statements for CyberOptic Corporation are in the Working Papers. The financial statements have been completed up to the trend analysis section. The following information is...
-
Recall the Martin-Beck Company distribution system problem in Section 11.3. a. Modify the formulation shown in Section 11.3 to account for the policy restriction that one plant, but not two, must be...
-
Henry is planning to purchase a Treasury bond with a coupon rate of 2.05% and face value of $100. The maturity date of the bond is 15 March 2033. (a)If Henry purchased this bond on 3 March 2020,what...
-
Kaitlin Carlton, a CPA sole practitioner, prepares tax returns each year for approximately 100 clients. Items 1 through 8 each represent an independent factual situation in which Kaitlin has prepared...
-
South Bridge Industries 2021 Cash Flow Statement (indirect method) Beginning cash $40,500 Net profit $8,532 Add depreciation $3,250 Accts receivable (increase)/Decrease ($19,000) Inventory...
-
If net operating income is $34,000, average operating assets are $204,000, and the minimum required rate of return is 15%, what is the residual income?
-
2. The device used to convert a binary number to a 7-segment display format is the a. multiplexer b. encoder decoder d. register 3. The binary number 11011101 is equal to the decimal number a. 121...
-
An online company is advertising a vacuum cleaner on sale for 55 % off the original price of $269.99. What is the sale price for the vacuum cleaner? Round your answer to the nearest cent.
-
NoDelay Delivery Corporation included the following disclosure note in an annual report: Stock-Based Compensation (in part) . . .Restricted shares of our common stock are awarded to key employees....
-
Bright Spark Trading has the following transactions in the last month of its year ending 3 1 December 2 0 2 2 . 9 Dec Collected in December 2 0 2 2 , $ 5 , 1 0 0 for service revenue earned and $ 5 0...
-
Question 3 of 3 rs Ariel received a loan of $35,000 at 3.5% compounded semi-annually. She had to make payments at the end of every half-year for a period of 6 years to settle the loan. a. Calculate...
-
According to a New York Times columnist, The estate tax affects a surprisingly small number of people. In 2003, . . . just 1.25 percent of all deaths resulted in taxable estates, with most of them...
-
If the following half-cells are the right-hand electrode in a galvanic cell with a standard hydrogen electrode on the left, calculate the cell potential. If the cell were shorted, indicate whether...
-
Find the mean and median of each of the following sets of data. Determine the deviation from the mean for each data point within the sets and find the mean deviation for each set. Use a spreadsheet...
-
Find the number of Na1 ions in 2.92 g of Na3PO4?
-
Which of the following is a fraud in which later payments on account are used to pay off earlier payments that were stolen? a. lapping b. kiting c. Ponzi scheme d. salami technique
-
Public charities are strictly prohibited from engaging in political campaigns for or against a candidate. Why do you think this is the case?
-
If the media contacts a board member in reference to the organization, what should her response be?

Study smarter with the SolutionInn App



