1. Hydrogen peroxide reacts with thiosulfate ion in slightlyacidic solution as follows: H 2 O 2 +...
Question:
1. Hydrogen peroxide reacts with thiosulfate ion in slightlyacidic solution as follows:
H2O2 + 2S2O32?+ 2 H+? 2H2O + S4O62?
The reaction rate is independent of the hydrogen ionconcentration in the pH range 4 to 6. The following data wereacquired at 25 ?C and pH 5.0. Initial concentrations:[H2O2] = 0.036 mol/L,[S2O32-] = 0.0204 mol/L
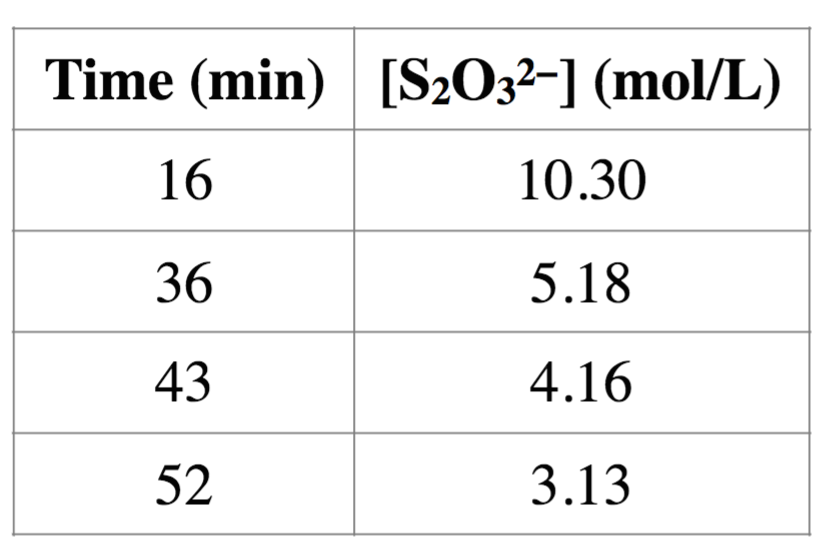
(a) What is the overall order of the reaction?
(b) What is the rate constant?
No information was given for [H2O2] for the course ofthe reaction
2. For the reaction, where M is an inert gas atom (such as He orAr),
O + NO + M ? NO2 + M
The rate constant exhibits the following temperaturebehavior:
at 300 K, k = 6 ? 109L2mol?2s?1; at 1000 K, k = 3 x1010 L2mol?2s?1.
Determine the activation energy and the prexponential factorusing the Arrhenius equation.

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris




