? ? ? 1. The powder X-ray diffraction patterns for 4 of the halide salts of Element
Fantastic news! We've Found the answer you've been seeking!
Question:

 ?
? ?
? ?
?
Transcribed Image Text:
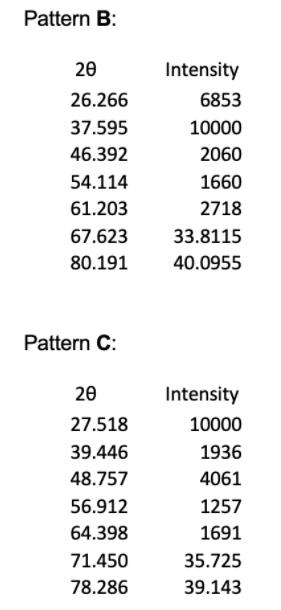
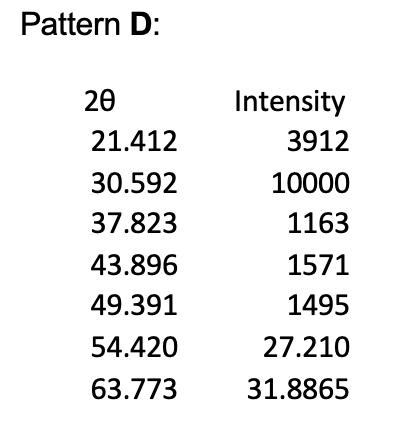
1. The powder X-ray diffraction patterns for 4 of the halide salts of Element X are shown below, and their first 5 peaks given in tables. Note that all patterns were obtained using Cu Ka radiation of wavelength = 1.54056 Å, and the halide and element X are in a 1:1 ratio. Pattern A 20 20.717 29.389 36.357 42.009 47.351 52.081 60.984 Intensity 556 10000 135 1714 165 26.0405 30.492 Pattern B: 20 26.266 37.595 46.392 54.114 61.203 67.623 80.191 Pattern C: 20 27.518 39.446 48.757 56.912 64.398 71.450 78.286 Intensity 6853 10000 2060 1660 2718 33.8115 40.0955 Intensity 10000 1936 4061 1257 1691 35.725 39.143 Pattern D: 20 21.412 30.592 37.823 43.896 49.391 54.420 63.773 Intensity 3912 10000 1163 1571 1495 27.210 31.8865 (e) (i) Given the ionic radius of element X is 181 pm, calculate the ionic radius of the halide in pattern C. (4 marks) (ii) In terms of Element X, assign chemical formulae to XRD patterns A to D. 1. The powder X-ray diffraction patterns for 4 of the halide salts of Element X are shown below, and their first 5 peaks given in tables. Note that all patterns were obtained using Cu Ka radiation of wavelength = 1.54056 Å, and the halide and element X are in a 1:1 ratio. Pattern A 20 20.717 29.389 36.357 42.009 47.351 52.081 60.984 Intensity 556 10000 135 1714 165 26.0405 30.492 Pattern B: 20 26.266 37.595 46.392 54.114 61.203 67.623 80.191 Pattern C: 20 27.518 39.446 48.757 56.912 64.398 71.450 78.286 Intensity 6853 10000 2060 1660 2718 33.8115 40.0955 Intensity 10000 1936 4061 1257 1691 35.725 39.143 Pattern D: 20 21.412 30.592 37.823 43.896 49.391 54.420 63.773 Intensity 3912 10000 1163 1571 1495 27.210 31.8865 (e) (i) Given the ionic radius of element X is 181 pm, calculate the ionic radius of the halide in pattern C. (4 marks) (ii) In terms of Element X, assign chemical formulae to XRD patterns A to D.
Expert Answer:

Related Book For 

Modern Portfolio Theory and Investment Analysis
ISBN: 978-1118469941
9th edition
Authors: Edwin Elton, Martin Gruber, Stephen Brown, William Goetzmann
Posted Date:
Students also viewed these chemistry questions
-
T-accounts for JCB Industries, Inc., are shown below and on the following page. Required: 1. Analyze these accounts and detail the appropriate journal entries that must have been made by JCB...
-
Selected accounts of Koffman Company are shown below and on the next page. Instructions After analyzing the accounts, journalize (a) The July transactions (b) The adjusting entries that were made on...
-
A narrow beam of X-ray radiation of wavelength 62 pm penetrates an aluminum screen 2.6 cm thick. How thick must a lead screen be to attenuate the beam just as much? The mass absorption coefficients...
-
Calculate the Total Cost of Ownership of the following 2 models and determine which model is a better buy. Model X Model Y $50,000 $60,000 Purchase price = Expected useful life = Maintenance cost per...
-
Who should be responsible for evaluating the greeting card merchandiser's performance?
-
The turbine shown in Fig. P5.114 develops \(100 \mathrm{hp}\) when the flowrate of water is \(20 \mathrm{ft}^{3} / \mathrm{s}\). If all losses are negligible, determine (a) the elevation \(h\), (b)...
-
Explain the difference between an asset and an expense in conventional financial accounting. Are pollution permits an asset? Comment on the view that letting companies hold pollution allowances as...
-
Analyzing and Interpreting Return on Assets Tiffany & Co. is one of the worlds premier jewelers and a designer of other fine gifts and housewares. Presented here are selected income statement and...
-
Forward Contracts and No-Arbitrage Pricing (20 Points) Under no-arbitrage pricing it follows that future price of a stock index corresponds to the following geometric Brownian motion (GBM): ST = Stx...
-
1. a. How much did the materials cost per unit change between June and July? b. How much did the conversion cost per unit change between June and July? 2. Which is the optimal choice of material and...
-
1- They share in common that they have anaplerotic and amphibole pathways with amino acids: a) Kinase b) Gluconeogenesis c) Hypoxia d) Lactate e) Fructose 6-phosphate to fructose 1,6-bisphosphate f)...
-
Len is asked to design a small water pump for an aquarium. The pump should deliver \(18.0 \mathrm{Lpm}\) of water at a net head of \(1.6 \mathrm{~m}\) at its best efficiency point. A motor that spins...
-
Why is it important to create a final project report, presentation, and lessons-learned report?
-
The following actions by a CPA may involve violations of the Rules of Conduct: 1. Agrees to be the committee chairperson for a local fund-raising activity. 2. Serves as an executor and trustee of the...
-
What type of information do you see in the Earned Value table?
-
What are the conditions associated with liability under Rule \(10 \mathrm{~b}-5\) of the 1934 Act?
-
3) Write a Fortran program that displays an amortization schedule. The program should read the loan amount, annual interest rate, and the loan term in months (from a single line). The formula for...
-
Show that the block upper triangular matrix A in Example 5 is invertible if and only if both A 11 and A 22 are invertible. Data from in Example 5 EXAMPLE 5 A matrix of the form A = [ A11 A12 0 A22 is...
-
Consider the purchase of a combination of two puts and a call. Assume that the call costs $5, the put costs $6, and the exercise price for the put or call is $50. Plot the profit versus the stock...
-
Consider the CAPM line shown below. What is the excess return of the market over the risk-free rate? What is the risk-free rate? h-0.04 +0.106,
-
Given the following situation: Draw the minimum variance curve and efficient frontier in expected return standard deviation space. Be sure to give the coordinates of all key points. Draw the security...
-
Meagan Casey, the sole proprietor and owner of Sierra Marina, would now like you to help her prepare some basic financial reports related to the companys accounts receivable and payable....
-
Creating More Supporting Reports for Larrys Landscaping & Garden Supply Scott wants you to help him provide more supporting reports. Create the reports he has requested, and write down the answers to...
-
Ocean View Flowers is in the wholesale distribution and sales industry and is located at 100 Ocean Ave. in Lompoc, CA 93436. Ocean View started business as a corporation on January 1, 2016, and owner...

Study smarter with the SolutionInn App


