1. Which of the following statements will explain why the surface tension of CHBr3 is greater than...
Fantastic news! We've Found the answer you've been seeking!
Question:
1. Which of the following statements will explain why the surface tension of CHBr3 is greater than CHCl3?
A. CHBr3 has a higher molar mass, more polarizable thus stronger dispersion forces.
B. CHCl3 exhibits dipole- dipole force thus lesser surface tension.
C. CHBr3 is less polarizable thus stronger dispersion force.
D. Chlorine is more electronegative than bromine thus CHBr3 has stronger H-bond than CHCl3.
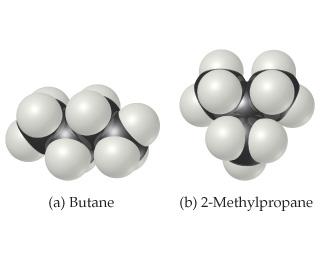
2. Why butane has a higher boiling point than 2-methylpropane, whose space-filling models are shown?
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:




