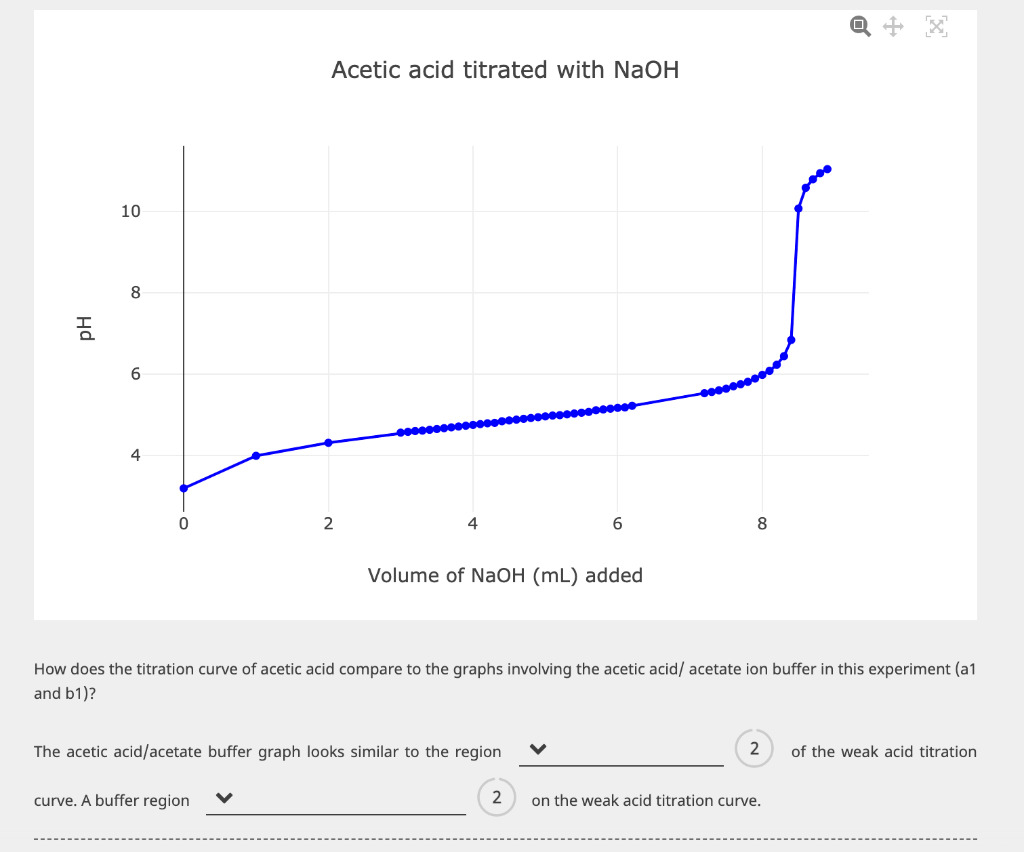
10 8 01 6 4 0 Acetic acid titrated with NaOH 2 curve. A buffer region...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
рп 10 8 01 6 4 0 Acetic acid titrated with NaOH 2 curve. A buffer region Volume of NaOH (mL) added The acetic acid/acetate buffer graph looks similar to the region 6 How does the titration curve of acetic acid compare to the graphs involving the acetic acid/ acetate ion buffer in this experiment (a1 and b1)? 2 8 2 on the weak acid titration curve. of the weak acid titration рп 10 8 01 6 4 0 Acetic acid titrated with NaOH 2 curve. A buffer region Volume of NaOH (mL) added The acetic acid/acetate buffer graph looks similar to the region 6 How does the titration curve of acetic acid compare to the graphs involving the acetic acid/ acetate ion buffer in this experiment (a1 and b1)? 2 8 2 on the weak acid titration curve. of the weak acid titration
Expert Answer:
Answer rating: 100% (QA)
16 5 In the aletic acid Vs NaOH Titation curve Buffer region is at pH 376 to 576 shown ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
A titration curve for NaCO3 titrated with HCl is shown here. Suppose that both phenolphthalein and bromocresol green are present in the titration solution. State what colors you expect to observe at...
-
The figure compares the titration of a monoprotic weak acid with a monoprotic weak base and the titration of a diprotic acid with strong base. (a) Write the reaction between the weak acid and the...
-
A weak acid HA (pKa 5.00) was titrated with 1.00 M KOH. The acid solution had a volume of 100.0 mL and a molarity of 0.100 M. Find the pH at the following volumes of base added and make a graph of pH...
-
In determining an employee's net pay, which of the following taxes would be deducted? a. FUTA taxes b. SUTA taxes c. FICA taxes d. All of these choices are correct.
-
Crafts Galore, a distributor of handmade gifts, operates out of owner Jenny Finns house. At the end of the current period, Jenny reports she has 1,500 units (products) in her basement, 30 of which...
-
Levels of carbon dioxide (CO 2 ) in the atmosphere are rising rapidly, far above any levels ever before recorded. Levels were around 278 parts per million in 1800, before the Industrial Age, and had...
-
Arthur Shore practiced accounting with a partnership for five years. Recently he opened his own accounting firm, which he operates as a professional corporation. The name of the new entity is Arthur...
-
Zeidler Company bought a building and the land on which the building is located for a total cash price of $ 356,000. The company paid transfer costs of $ 4,000. Renovation costs on the building were...
-
Price-earnings ratio (PE ratio) is a salient statistic about a stock. In valuation multiple approach, what are the potential issues of finding comparable firms based on PE ratio?
-
Suppose that there are p = 4 quality characteristics, and in correlation form all four variables have variance unity and that all pairwise correlation coefficients are 0.9. The in-control value of...
-
Locate the centroid y the composite body shown in the figure. XG. = ? 4cm 2cm Ticm [1cm X 14 cm 2cm
-
1. Listening is one way to improve communication in negotiation. What are the three major forms of Listening? 2. The five major sources of power as outlined by the text include. 3. What are the...
-
Susan's mother is currently living in a nursing home, and she is worried about the financial drain of her mother's long-term care costs. She would like to start planning for her own potential future...
-
1. Has anyone thought that it is conceivable that an organization with a self-assured income can in any case wind up in desperate waterways? 2. Would you be able to characterize the significance of...
-
Access the simulation Finding and Leveraging Voice (ELL) 2. Complete the simulation, taking note of what actions/decisions were successful and which actions/decisions lead to suggestions or other...
-
A popular app, Zolo, has come on the market for streaming pet-friendly music while others in the household are away from home for work or errands. Every month, the app attracts 60% of non-subscribing...
-
For this code you must use the 'ready to program' ide in java, and do not use scanner class. Ready to program ide: https://ready-to-program-with-java.software.informer.com/1.7/ I Write a program that...
-
QUESTION 2 The CEO of Farisha Hijab Sdn Bhd insisted on further investigation to be carried out that he also required Mr Muaz to conduct the analysis of variance for the material and labour of the...
-
Of the seven three-dimensional primitive lattices, (a) Which one has a unit cell where the a and b lattice vectors form abase that is an arbitrary parallelogram (like the unit cell of a...
-
A 0.100 Msolution of bromoacetic acid (BrCH2COOH) is 13.2% ionized. Calculate [H+], [BrCH2COOH] and Ka for bromoacetic acid.
-
A certain AB4 molecule has a "seesaw" shape: From which of the fundamental geometries shown in Figure 9.3 could you remove one or more atoms to create a molecule having this seesaw shape? 109.5 180...
-
For each of the following events concerning disclosure of events that took place after year end, discuss the manner in which it should be disclosed in the financial statements or the audit report....
-
Determining whether a subsequent event requires an adjustment to the financial statements is difficult in many circumstances. Discuss the primary difference between subsequent events that require...
-
Each of the following techniques for managing earnings was described in the chapter: - "Big Bath" charges - Write-off of acquired assets - "Cookie Jar" reserves - Abuse of materiality - Questionable...

Study smarter with the SolutionInn App


