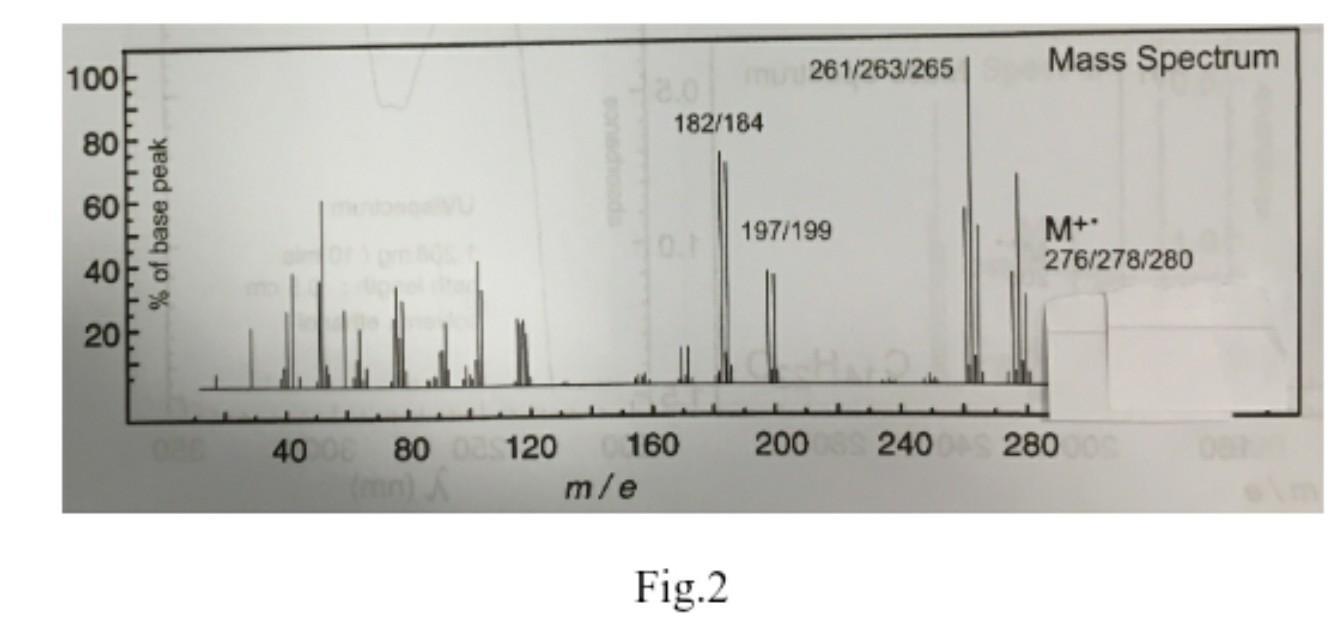
4. Reactivity considerations suggest that an unknown compound, which has the mass spectrum shown in Fig....
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
4. Reactivity considerations suggest that an unknown compound, which has the mass spectrum shown in Fig. 2, is a vinyl halide. Combustion analysis gave the following results: Carbon, 38.89 %; Hydrogen, 3.63 %. In order to finish the combustion analysis properly, the operator needs to know which halide to test for. Which halide would you suggest? Postulate a molecular formula for this unknown compound. Explanation of the fragmentation pattern is not required! 100 80 60 40 20 % of base peak 40 00 80 (mn) 8.0 182/184 10.1 120 0160 m/e mu261/263/265 Fig.2 197/199 200 240 Mass Spectrum M+ 276/278/280 280000 4. Reactivity considerations suggest that an unknown compound, which has the mass spectrum shown in Fig. 2, is a vinyl halide. Combustion analysis gave the following results: Carbon, 38.89 %; Hydrogen, 3.63 %. In order to finish the combustion analysis properly, the operator needs to know which halide to test for. Which halide would you suggest? Postulate a molecular formula for this unknown compound. Explanation of the fragmentation pattern is not required! 100 80 60 40 20 % of base peak 40 00 80 (mn) 8.0 182/184 10.1 120 0160 m/e mu261/263/265 Fig.2 197/199 200 240 Mass Spectrum M+ 276/278/280 280000
Expert Answer:
Answer rating: 100% (QA)
4 Based on the reactivity considerations it is likely that the unknown compound is a vinyl halide Th... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The Avemar ferry, shown in Figure PI0.9(a), is a large 670-ton ferry hydrofoil built for Mediterranean ferry service. It is capable of 45 knots (52 mph) [29]. The boat's appearance, like its...
-
The structural representation shown in the margin is a Newman projection of the conformer of butane that is (a) Gauche eclipsed (b) Anti gauche (c) Anti staggered (d) Anti eclipsed
-
The gas-turbine cycle shown in Fig P11.54 is used as an automotive engine. In the first turbine, the gas expands to pressure P5, just low enough for this turbine to drive the compressor. The gas is...
-
The graph of g consists of two straight lines and a semicircle. Use it to evaluate each integral. (a) g(x) dr (b) g(x) dr (c) () di y=g(x)
-
Retail chains want to ensure that every time a customer returns to purchase something, the record of that purchase can be matched with previous data of that customer. What objects that consumers...
-
A researcher studying the properties of ions in the upper atmosphere wishes to construct an apparatus with the following characteristics: Using an electric field, a beam of ions, each having charge...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
The following transactions were completed by Navarez Inc., whose fiscal year is the calendar year: 2014 July 1. Issued $88,000,000 of 10-year, 10% callable bonds dated July 1, 2014, at a market...
-
You are a co-op student at Modus Biosystems and have been asked to research whether there are ways to save money on their legal liability insurance. Modus currently pays $75,000 a year for a policy...
-
Variable and fixed costs, profitability of order, opportunity cost Healthy Hearth specializes in lunches for health-conscious people. The company produces a small selection of lunch offerings each...
-
A married couple has $65,000 of income in 2019. They file a joint return. The first spouse is 70years old, while the second is 61years old. What is their standard deduction?
-
Cepeda Corporation has $300,000 that it wants to invest. It is choosing between AT&T corporate bonds that yield 6.3 percent, state of Ohio municipal bonds that yield 4.00 percent, and AT&T preferred...
-
JLM Services was organized on August 1, 20Y5. A summary of the revenue and expense transactions for August are as follows: Line Item Description Amount Fees earned $690,400 Wages expense 352,100...
-
For a business to build customer value, it must manage customer relationships through a model called IDIC. What does IDIC stand for? Individual, Differentiate, Insight and Customer Individual,...
-
If Susan sues Will and Frank's Furniture for $ 8 0 , 0 0 0 in damages resulting from her injuries in federal district court in Montpelier, Vermont, will the court have proper subject matter...
-
If you are trying to forecast your company's growth for the next year, what should be your primary consideration? the company's leverage the company's owner equity the company's recent performance...
-
Graph the system of inequalities. x2 y25-2x *** Use the graphing tool to graph the system of inequalities. Click to enlarge graph The vertex(vertices) is (are) (Type an ordered pair. Use a comma to...
-
A bar of length = 1 has one fixed and one free end and stiffness function c(x) = 1 - x. Find the displacement when subjected to a unit force. Pay careful attention to the boundary condition at the...
-
Show that the (S,S) enantiomer of this (R,R) diastereomer of 1-bromo-1,2-diphenylpropane also undergoes E2 elimination to give the cis diastereomer of the product. (We do not expect these achiral...
-
Show how crossed Claisen condensations could be used to prepare the following esters. (a) (b) (c) (d) Ph C-CH-C OCH,CH CH CH-C-OCH3 C-C-oCH Eto C CH-C OCH,CH Ph CH,CH,CH,
-
Give the general classification of each compound. (a) glyceryl tripalmitate (b) (c) (d) (e) (f) CH3(CH210 CH2 Na CH3 (CH21C (CH216-CH3 tetradecyl octadecanoate CH3 CH HoC caryophyllene (from cloves)...
-
Several years ago, Catherines Cattery Supplies signed a contract to deliver 5000 units of a special product each year to one of its customers at a price of $15 per unit. This contract, which runs for...
-
Cairns Farming is a small family run dairy farm, which is finding it hard to continue in business. After visiting a farm in Nienberg, Germany, it is considering buying equipment that will enable it...
-
The Anderson Group has identified the following capital expenditure proposals as acceptable. Only $150 000 is available for investment. Required: Determine the combination of proposals to be...

Study smarter with the SolutionInn App


