11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
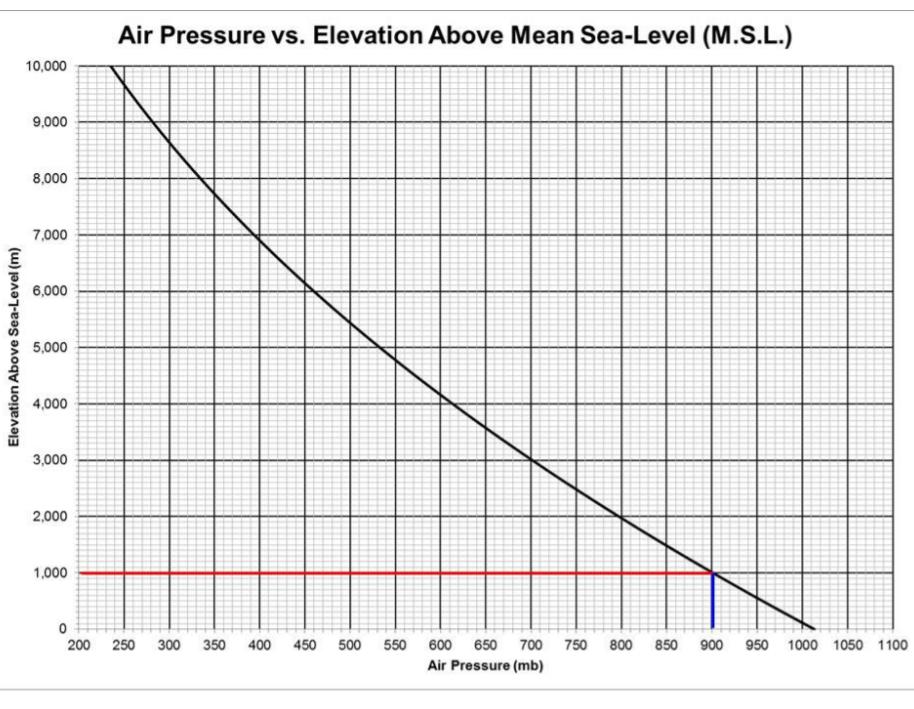
11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July.
Expert Answer:
Answer rating: 100% (QA)
11 We find that elevation is given in feet while the height in th... View the full answer

Related Book For 

A Survey of Mathematics with Applications
ISBN: 978-0134112107
10th edition
Authors: Allen R. Angel, Christine D. Abbott, Dennis Runde
Posted Date:
Students also viewed these law questions
-
The elevation E, in meters, above sea level at which the boiling point of water is t degrees Celsius is given by the function E(t) = 1000(100 - t) + 580(100 - t) 2. At what elevation is the boiling...
-
The summit of Mount Everest is 8850 m above sea level. (a) How much energy would a 90 kg climber expend against the gravitational force on him in climbing to the summit from sea level? (b) How many...
-
The elevation of a mountain above sea level at (x, y) is 3000e-(x2+2y2)/100 meters. The positive x-axis points east and the positive y-axis points north. A climber is directly above (10, 10). If the...
-
Prepare a statement of cash flows in proper form using the inflows and outflows from questions 4-15. Assume net income (earnings after taxes) from the 2018 income statement was $10,628, and $5,000 in...
-
Multinational transfer pricing causes serious concern for various corporate stakeholders . Identify potential concerns from the viewpoint of a. Minority owners of a foreign affiliate, b. Foreign...
-
A 5.0-g bullet with a speed of is fired horizontally into a 0.75-kg wooden block at rest on a table. If the block containing the bullet slides a distance of 0.20 m before coming to rest, (a) What is...
-
Using divisibility rules, determine if 87,762 is divisible by 10 .
-
Enos Printing Corp. uses a job order cost system. The following data summarize the operations related to the first quarters production. 1. Materials purchased on account $192,000, and factory wages...
-
As a credit manager, it is very important to evaluate the borrower before sanctioning the loan. Explain the four C's of traditional credit analysis. While preparing the report, you came to know abo...
-
Capstone Case: Sunrise Bakery Expansion The Sunrise Bakery Corporation was originally founded in Houston, TX in 1991 by Griffin Harris, who currently serves as the company's Chief Executive Officer....
-
The probability that a skeet shooter can hit 27 or more targets in 35 total shots is 2.2145414080299e-07. Find the probability of this shooter hitting the target when he shoots once at a single...
-
Briefly describe where the QA function should be placed in the organizational hierarchy of the information systems function.
-
Catskills Corporation guarantees its snowmobiles for three years. Company experience indicates that warranty costs will add up to 5% of sales. Assume that the Catskills dealer in Colorado Springs...
-
A \($400,000\) bond priced at 105.5 can be bought or sold for a. \($400,000\) + interest. b. $422,000. c. $22,000. d. $42,200
-
Why is QA training an important means of discernment for QA personnel in relation to QA standards and procedures?
-
Briefly explain the Japanese notion of Kaizen. Describe two principles on which Kaizen is based.
-
Using the below what is the best suited identification strategy? Research question - Does exposure to violent video games lead to an increase in aggressive behavior among adolescents? -For this...
-
Teasdale Inc. manufactures and sells commercial and residential security equipment. The comparative unclassified balance sheets for December 31, 2015 and 2014 are provided below. Selected missing...
-
Change the percent to a decimal number. 0.00045%
-
Use lattice multiplication to determine the product. 4 x 327
-
Evaluate the expression. 8 P 4
-
1.3 Isaac Dez of Brazil. Isaac Dez Peris lives in Rio de Janeiro, Brazil. While attending school in Spain, he meets Juan Carlos Cordero from Guatemala. Over the summer holiday, Isaac decides to visit...
-
1.4 Munich to Moscow. For your post-graduation cel- ebratory trip, you decide to travel from Munich, Germany, to Moscow, Russia. You leave Munich with 15,000 euros (EUR) in your wallet. Wanting to...
-
1.5 Moscow to Tokyo. After spending a week in Moscow, you get an email from your friend in Japan. He can get you a very good deal on a plane ticket and wants you to meet him in Tokyo next week to...

Study smarter with the SolutionInn App


