Show the product of reaction of the indicated ester with LDA, followed by butyl iodide. tho CH,CH3
Fantastic news! We've Found the answer you've been seeking!
Question:
Show the product of reaction of the indicated ester with LDA, followed by butyl iodide.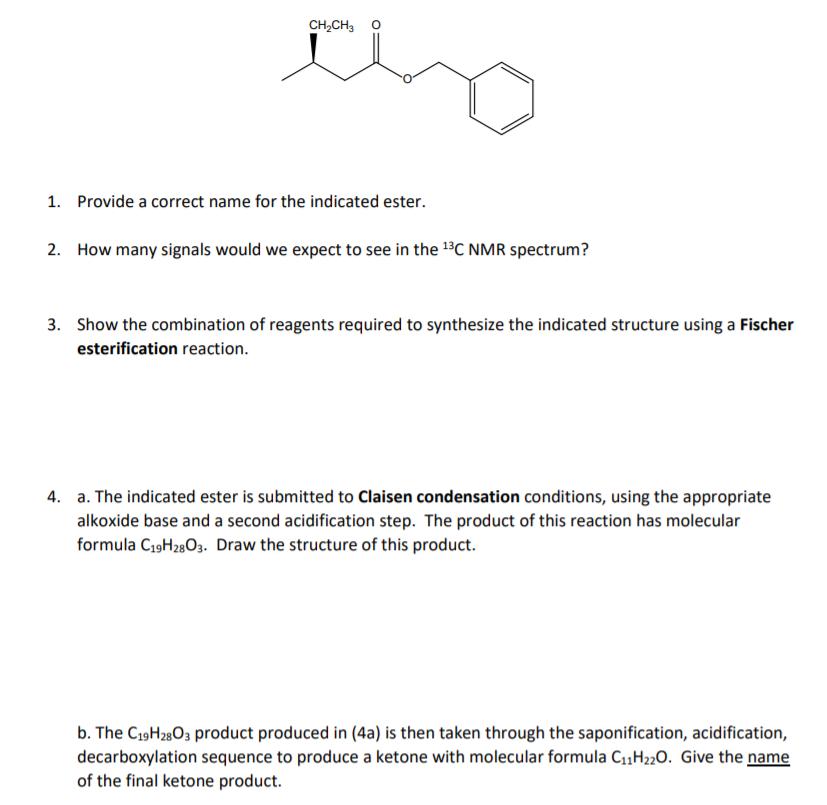
Transcribed Image Text:
tho CH,CH3 1. Provide a correct name for the indicated ester. 2. How many signals would we expect to see in the BC NMR spectrum? 3. Show the combination of reagents required to synthesize the indicated structure using a Fischer esterification reaction. 4. a. The indicated ester is submitted to Claisen condensation conditions, using the appropriate alkoxide base and a second acidification step. The product of this reaction has molecular formula C19H2803. Draw the structure of this product. b. The C19H2803 product produced in (4a) is then taken through the saponification, acidification, decarboxylation sequence to produce a ketone with molecular formula C1,H220. Give the name of the final ketone product. tho CH,CH3 1. Provide a correct name for the indicated ester. 2. How many signals would we expect to see in the BC NMR spectrum? 3. Show the combination of reagents required to synthesize the indicated structure using a Fischer esterification reaction. 4. a. The indicated ester is submitted to Claisen condensation conditions, using the appropriate alkoxide base and a second acidification step. The product of this reaction has molecular formula C19H2803. Draw the structure of this product. b. The C19H2803 product produced in (4a) is then taken through the saponification, acidification, decarboxylation sequence to produce a ketone with molecular formula C1,H220. Give the name of the final ketone product.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The IR spectrum, 13C NMR spectrum, and 1HNMR spectrum of an unknown compound (C6H8O3) appear next. Determine the structure, and show how it is consistent with the spectra. wavelength (um) 5 5.5 6 710...
-
Show what esters would undergo Claisen condensation to give the following b-keto esters. (a) (b) (c) CH,CHCH,-C" - CH,CH,-CH-C-OCH,CH, Ph-CH,-C" O Ph CHCOCH CH(CH32
-
Draw the expected broadband-decoupled 13C NMR spectra of the following compounds. Use Figure 13-41 (page 603) to estimate the chemical shifts. (a) (b) (c) (d) C CH H,C CH C-H C-C
-
Students will present a draft IMC strategy for a start-up company and be asked to analyze its potential effectiveness, identifying gaps and making recommendations for improvement of the strategy....
-
Plymouthville leased equipment with a fair market value of $ 905,863. The life of the noncancellable lease is 10 years, and the economic life of the property is 10 years. Using an 8 percent interest...
-
The following data for Gridiron Industries, Inc., relate to the payroll for the week ended December 7, 2012: Employees Csonka and Motley are office staff, and all of the other employees are sales...
-
Celestial Inc. is organized into 3 distinct divisions. The company is currently contemplating the elimination of either Division 1 or Division 2 because they are showing a pre-tax loss. An annual...
-
Contrast the critical success factors (CSFs) and SWOT (i.e., strengths, weaknesses, opportunities, and threats) approaches for assessing opportunities as part of a strategic IS planning process....
-
Purpose of a Pre-Convention Meeting. Please give me a heads up on: Imagine that you are the Convention Services Manager for a major convention center - the Fort Worth Convention Center. You have a...
-
1 Leila Bannon, your supervisor and a fellow CPA, has been invited to participate in a symposium on ethics in the workplace. This event will bring together professionals from different fields who...
-
During exercise, your body produces sweat (basically water) which absorbs heat from the body to evaporate, thereby cooling it. Suppose that 31 mL of sweat evaporates every minute vou exercise. In 16...
-
A firm's cost of unlevered equity is 11% and its pretax cost of debt is 8%.The firm has a debt-equity ratio of 0.4.If the tax rate is 40%, what is the firm's cost of levered equity? A firm recently...
-
If there are 9 directors that need to be elected but their seats are staggered evenly over 3 years, how many shares do you need to own in order to guarantee a seat on the board under cumulative...
-
Asset 1 has a standard deviation of 0 . 1 0 and an expected return of 7 % . Asset 2 has a standard deviation of 0 . 1 6 and an expected return of 1 1 % . The covariance between them is 0 . 0 ....
-
What are the different types of Inventory Accounts? Why do we have to manage inventory? Who handle the inventory? What are the documents and forms to evidence inventory? 5. how are these inventory...
-
Q3: Define behavioural finance and how the psychology and neuroscience behind it has evolved over the past decade?
-
Here are some important figures from the budget of Nashville Nougats, Incorporated, for the second quarter of 2022: Credit sales Credit purchases Cash disbursements Wages, taxes, and expenses...
-
A number of years ago the United Food and Commercial Workers Union organized 800 workers of the 1035 employees at one of the Wilson Brothers food operations in Toronto, Ontario. The employees include...
-
The reactions shown will all be encountered in Chapter 6. Classify each according to whether it proceeds by oxidation of carbon, by reduction of carbon, or by a process other than...
-
(Z)-9-Tricosene [(Z)-CH3(CH2)7CHCH(CH2)12CH3] is the sex pheromone of the female housefly. Synthetic (Z)-9-tricosene is used as bait to lure male flies to traps that contain insecticide. Using...
-
On the basis of the mechanism of cationic polymerization, predict the alkenes of molecular formula C12H24 that can most reasonably be formed when 2-methylpropene [(CH3)2C=CH2] is treated with...
-
When constructing a common-sized income statement, all amounts are expressed as a percentage of: a. net income. b. gross profit. c. net sales. d. income from operations.
-
The current ratio of Hydro Company at December 31, 2018, calculated using the above data, was 3.13 and the company's working capital was \(\$ 170,000\). Which of the following would happen if the...
-
What was the firm's inventory turnover for 2018? a. 6.67 b. 4 c. 6 d. 3.69 Sales revenues for 2018 were \(\$ 800,000\), gross profit was \(\$ 320,000\), and net income was \(\$ 36,000\). The income...

Study smarter with the SolutionInn App



