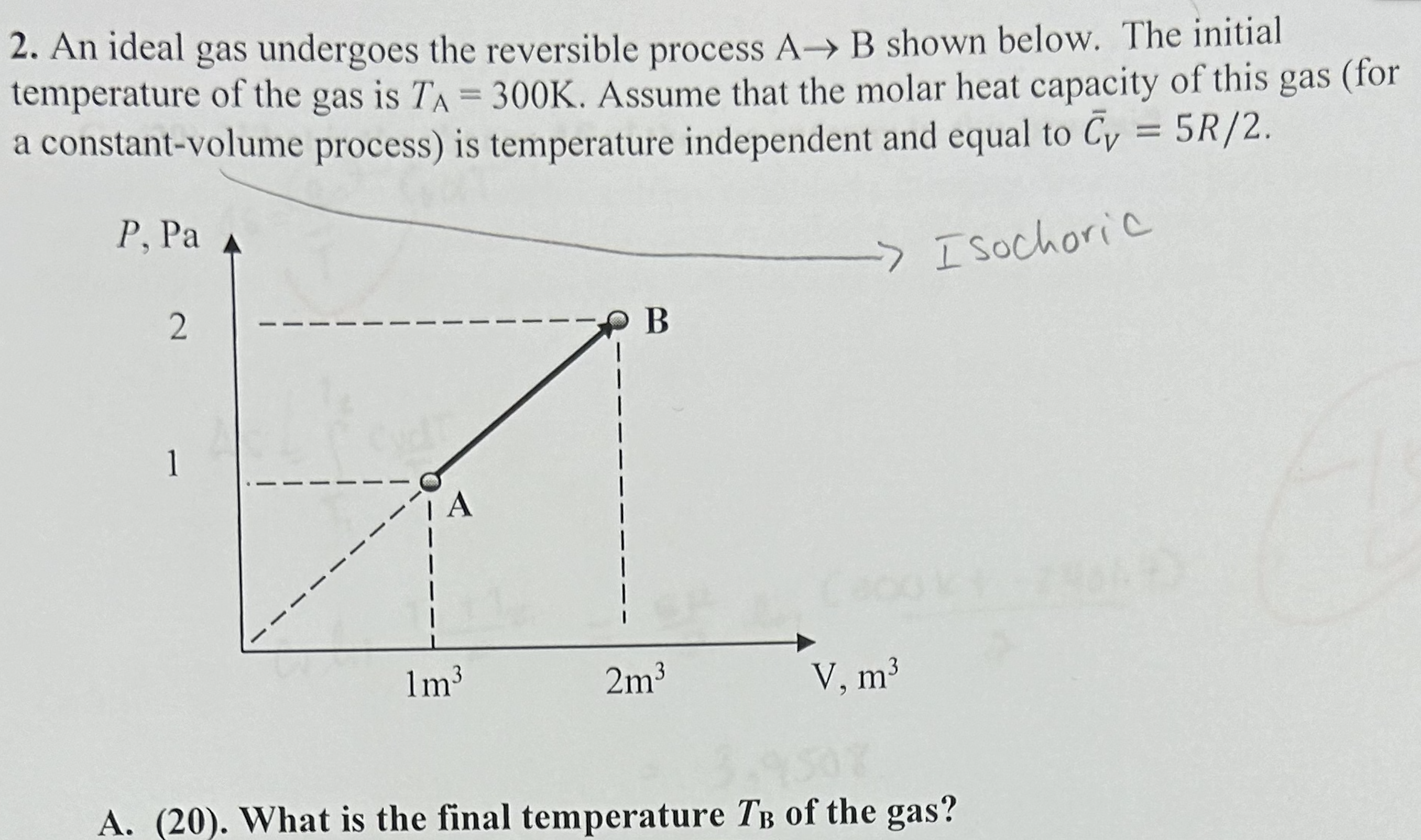
2. An ideal gas undergoes the reversible process A B shown below. The initial temperature of...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
2. An ideal gas undergoes the reversible process A B shown below. The initial temperature of the gas is TA = 300K. Assume that the molar heat capacity of this gas (for a constant-volume process) is temperature independent and equal to Cy = 5R/2. P, Pa 2 -> Isochoric B 1 1m 2m V, m 9507 A. (20). What is the final temperature TB of the gas? 4. A vessel contains n moles of an ideal gas 1 and 2 moles of ideal gas 2 separated by a wall. Both gases are at the same pressure P and temperature T equal to the temperature of the surroundings. The vessel is not thermally insulated from the surroundings. T n2 P, T P, T A. (35) Calculate the change AS of the entropy of the gases after the wall is removed and the gases are mixed. 3(50). A cup of hot coffee at T = 360K is brought to the classroom, whose temperature is T = 300K. The heat capacity of the coffee in the cup is Cp = 400J/K, and the heat capacity of the cup itself is negligible. Estimate the change of the entropy of the Universe resulting from the coffee getting cold and attaining the room temperature. For the purpose of this problem the Universe = coffee + surroundings, where you can think of the surroundings as a giant body (of infinitely large heat capacity) at T = T. 2. 5* (70 extra credit points). A balloon filled with hot air at 7=400K and P = 1 atm is released at sea level and rises until it stops h meters above sea level. The balloon is made from a weightless, rigid material that does not conduct heat such that the volume of the balloon remains constant, and there is no the heat exchange between the balloon and the surroundings. The atmosphere is an ideal gas whose temperature is constant and equal to T = 300K at all elevations. Also assume that the atmospheric pressure changes with the elevation h above sea level according to the barometric formula (derived in the class): P=P exp(-Mgh/RT), where Po= 1 atm and the average molar mass of air is M = 29 g/mol. (Yes, this is the balloon from HW1). Estimate the change of the entropy of the surroundings (i.e., the atmosphere) as a result of this process (i.e. after the balloon stops rising). 2. An ideal gas undergoes the reversible process A B shown below. The initial temperature of the gas is TA = 300K. Assume that the molar heat capacity of this gas (for a constant-volume process) is temperature independent and equal to Cy = 5R/2. P, Pa 2 -> Isochoric B 1 1m 2m V, m 9507 A. (20). What is the final temperature TB of the gas? 4. A vessel contains n moles of an ideal gas 1 and 2 moles of ideal gas 2 separated by a wall. Both gases are at the same pressure P and temperature T equal to the temperature of the surroundings. The vessel is not thermally insulated from the surroundings. T n2 P, T P, T A. (35) Calculate the change AS of the entropy of the gases after the wall is removed and the gases are mixed. 3(50). A cup of hot coffee at T = 360K is brought to the classroom, whose temperature is T = 300K. The heat capacity of the coffee in the cup is Cp = 400J/K, and the heat capacity of the cup itself is negligible. Estimate the change of the entropy of the Universe resulting from the coffee getting cold and attaining the room temperature. For the purpose of this problem the Universe = coffee + surroundings, where you can think of the surroundings as a giant body (of infinitely large heat capacity) at T = T. 2. 5* (70 extra credit points). A balloon filled with hot air at 7=400K and P = 1 atm is released at sea level and rises until it stops h meters above sea level. The balloon is made from a weightless, rigid material that does not conduct heat such that the volume of the balloon remains constant, and there is no the heat exchange between the balloon and the surroundings. The atmosphere is an ideal gas whose temperature is constant and equal to T = 300K at all elevations. Also assume that the atmospheric pressure changes with the elevation h above sea level according to the barometric formula (derived in the class): P=P exp(-Mgh/RT), where Po= 1 atm and the average molar mass of air is M = 29 g/mol. (Yes, this is the balloon from HW1). Estimate the change of the entropy of the surroundings (i.e., the atmosphere) as a result of this process (i.e. after the balloon stops rising).
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
You are considering a job offer. The job offers an annual salary of $42,000, $45,000, and $48,000 a year for the next three years, respectively. The offer also includes a starting bonus of $1,000...
-
Specify the minimum type(s) of operation(s) the database must support (remote request, remote transaction, distributed transaction, or distributed request) to perform the following operations: At...
-
Determine whether there exists a constant c such that the line x + cy = 1: (a) Has slope 4 (b) Passes through (3, 1) (c) Is horizontal (d) Is vertical
-
What are the two ways a party may obtain a voluntary dismissal of an action?
-
Collins Corporation purchased office equipment at the beginning of 2009 and capitalized a cost of $2,000,000. This cost figure included the following expenditures: The company estimated an eight-year...
-
What groups of individuals might be more responsive to the increase on preservation age?
-
Write a program to place ten queens on 10 x 10 chessboard in such a way that one queen is to be in each row. A program will use 2 DIMENIONAL array x[r][c] to do this configuration. If x[r] has value...
-
explores the day-to-day issues that supply chain managers deal with to ensure that supply chains accomplish their purpose in creating and delivering goods to customers. How do supply chains relate...
-
The Gourmand Cooking School runs short cooking courses a small campus. Management has identified two cost drivers that it uses in its budgeting and performance reports the number of courses and the...
-
Paul Havlik promised his grandson Jamie that he would give him $6,400 8 years from today for graduating from high school. Assume money is worth 8% interest compounded semiannually. What is the...
-
What are the HIPAA - related issues surrounding the use of social media?
-
Calculate Social Security and Medicare tax for the below - listed employees of TCLH Industries, a manufacturer of cleaning products. None of the employees files as married filing separately on their...
-
Natural-language makes more sense in a mobile environment than in a desktop environment because the users of mobile devices are on the go and want to use their hands as little as possible. NLP adds a...
-
Dr. Chan obtained a $15,000 demand loan at prime plus 1.5% on September 13 from the Bank of Montreal to purchase a new dental X-ray machine. Fixed payments of $700 will be deducted from the dentists...
-
As the final edits to the Sixth Edition were being made, the coronavirus pandemic struck and it was decided to expand Problem P9-7 and apply it to the pandemic. (Mayur Tikmani, Devosmita Sen, Manjeet...
-
Styrene can be produced from ethylbenzene by the following reaction: ethylbenzenestyrene+H2(1) However, several irreversible side reactions also occur: ethylbenzene benzene + ethylene (2)...
-
The elementary gas-phase reaction A + B 2C in Problem P11-8B is now continued and carried out in packed-bed reactor. The entering molar flow rates are F A0 = 5 mol/s, F B0 = 2F A0 , and F I = 2F A0...
-
During 2020, Many Miles Trucking paid salary expense of \(\$ 40\) million. At December 31, Many Miles accrued salary expense of \(\$ 2\) million. Many Miles paid \(\$ 1.9\) million to its employees...
-
Answer the following questions about prepaid expenses: a. On November 1, World Travel Ltd. prepaid \(\$ 6,000\) for three months' rent. Give the adjusting entry to record rent expense at December 31....
-
Ford Canada sells large fleets of vehicles to auto rental companies, such as Budget and Avis. Suppose Budget is negotiating a contract with Ford to purchase 1,000 Explorers by the end of next year....

Study smarter with the SolutionInn App


