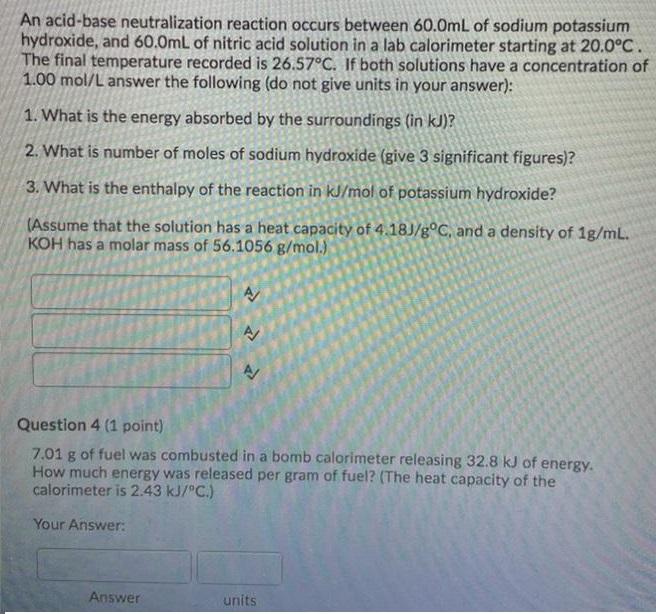
An acid-base neutralization reaction occurs between 60.0mL of sodium potassium hydroxide, and 60.0mL of nitric acid...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
An acid-base neutralization reaction occurs between 60.0mL of sodium potassium hydroxide, and 60.0mL of nitric acid solution in a lab calorimeter starting at 20.0°C. The final temperature recorded is 26.57°C. If both solutions have a concentration of 1.00 mol/L answer the following (do not give units in your answer): 1. What is the energy absorbed by the surroundings (in kJ)? 2. What is number of moles of sodium hydroxide (give 3 significant figures)? 3. What is the enthalpy of the reaction in kJ/mol of potassium hydroxide? (Assume that the solution has a heat capacity of 4.18J/g°C, and a density of 1g/mL. KOH has a molar mass of 56.1056 g/mol.) Question 4 (1 point) 7.01 g of fuel was combusted in a bomb calorimeter releasing 32.8 kJ of energy. How much energy was released per gram of fuel? (The heat capacity of the calorimeter is 2.43 kJ/"C.) Your Answer: Answer units An acid-base neutralization reaction occurs between 60.0mL of sodium potassium hydroxide, and 60.0mL of nitric acid solution in a lab calorimeter starting at 20.0°C. The final temperature recorded is 26.57°C. If both solutions have a concentration of 1.00 mol/L answer the following (do not give units in your answer): 1. What is the energy absorbed by the surroundings (in kJ)? 2. What is number of moles of sodium hydroxide (give 3 significant figures)? 3. What is the enthalpy of the reaction in kJ/mol of potassium hydroxide? (Assume that the solution has a heat capacity of 4.18J/g°C, and a density of 1g/mL. KOH has a molar mass of 56.1056 g/mol.) Question 4 (1 point) 7.01 g of fuel was combusted in a bomb calorimeter releasing 32.8 kJ of energy. How much energy was released per gram of fuel? (The heat capacity of the calorimeter is 2.43 kJ/"C.) Your Answer: Answer units
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The heat capacity of a bomb calorimeter was determined by burning 6.79 g of methane (energy of combustion = 802 kJ/ mol CH4) in the bomb. The temperature changed by 10.8oC. a. What is the heat...
-
An ideal gas has a molar heat capacity Cv at constant volume. Find the molar heat capacity of this gas as a function of its volume V, if the gas undergoes the following process: (a) T = Toe v ; (b) p...
-
An aqueous acetic-acid solution containing 6.0 moles of acid per liter is to be extracted in the laboratory with chloroform at 25oC to recover the acid (B) from chloroform-insoluble impurities...
-
What is the output of the following? A. 1223445 B. 2445 C. 22445 D. 223445 E. 2233445 F. None of the above. public class InitOrder { } {System.out.print("1"); } static System.out.print("2"); } public...
-
In the 1998 Minnesota gubernatorial election, there were three main candidates: Norm Coleman (the Republican), Jesse "The Body" Ventura (an Independent), and Hubert Humphrey (the Democrat). Although...
-
Refer to the information for Speedy Pete's above. Speedy Pete's controller wants to calculate the fixed and variable costs associated with its cutting-edge delivery service. Speedy Pete's is a small...
-
What is the relationship between globalization and nationalism in the current-day world?
-
Floyd Corporation had the following transactions pertaining to debt investments. Jan. 1 Purchased 50 8%, $1,000 Petal Co. bonds for $50,000 cash plus brokerage fees of $900. Interest is payable...
-
A pendulum inside a moving car is seen to make an angle of 30 o with the vertical for a duration of 20s. Calculate the change in velocity of the car in this duration?
-
How would the legal realists (e.g., Karl Llewellyn and Oliver Wendell Holmes) view the current relationship between social psychology and the law?
-
Imagine you are an audit manager for Ali Timam and partners - an audit firm based in Muscat. You have just been asked by one of your existing clients, Zubair corporation, for help in compliance with...
-
Assume in a given month, Japan's export to the U.S. increased. How such an increase will affect the Japanese Yen? From a U.S. perspective, how this increase will affect the U.S. dollar? Knowing that...
-
Explain three different reasons as to why the national debt may be a burden on current and future generations. Explain whether these reasons apply to the US now.
-
It would assist us if you would identify whether there are any network effects in the decisions of cable customers to "cut the cord" and networks to provide OTT programming. If more networks sell...
-
Consider two twins 25 years old. One twin steps on a spaceship (after studying physics in college) and travels through space at 0.95 c for 4 years according to her. How old is each twin when she...
-
Which combination of investments would likely be most suitable for an investor who is willing to accept a potential loss of investment capital to achieve a high growth rate?
-
The face value of the note is $ 3 3 7 million. Assume the 5 . 3 7 5 % notes due October 1 , 2 0 2 2 , were originally issued on October 1 , 2 0 1 6 , at 9 9 . 5 . Tina Inc. incurred $ 1 4 6 , 0 0 0...
-
Refer to the information from Exercise 22-19. Use the information to determine the (1) Weighted average contribution margin , (2) Break-even point in units, and (3) Number of units of each product...
-
Determine the molecular formula of a saturated acyclic hydrocarbon with an M peak at m/z = 100 with a relative intensity of 27.32%, and an M + 1 peak with a relative intensity of 2.10%.
-
The UV spectra of tryptophan, tyrosine, and phenylalanine are shown here. Each spectrum is that of a 1 Ã 10-3 solution of the amino acid, buffered at Calculate the approximate molar...
-
Use the symbols δ+ and δ- to show the direction of polarity of the indicated bond in each of the following compounds (for example, a. HO--H b. F--Br c. H3C--NH2 d. H3C--Cl...
-
The rotor shown in Fig. 9.44 (a) is balanced temporarily in a balancing machine by adding the masses \(m_{1}=m_{2}=90 \mathrm{~g}\) in the plane \(A\) and \(m_{3}=m_{4}=90 \mathrm{~g}\) in the plane...
-
A turbine rotor is run at the natural frequency of the system. A stroboscope indicates that the maximum displacement of the rotor occurs at an angle \(229^{\circ}\) in the direction of rotation. At...
-
The cylinders of a four-cylinder in-line engine are placed at intervals of \(300 \mathrm{~mm}\) in the axial direction. The cranks have the same length, \(100 \mathrm{~mm}\), and their angular...

Study smarter with the SolutionInn App


