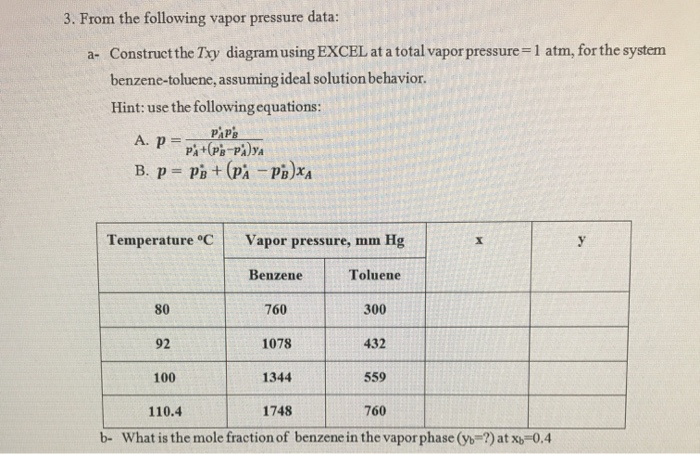
3. From the following vapor pressure data: a- Construct the Txy diagram using EXCEL at a...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
3. From the following vapor pressure data: a- Construct the Txy diagram using EXCEL at a total vapor pressure=1 atm, for the system benzene-toluene, assuming ideal solution behavior. Hint: use the following equations: PAP'S A. P= PA+ (PB-PAYA B. P= PB + (PA-PB)XA Temperature °C 80 92 100 Vapor pressure, mm Hg Benzene 760 1078 1344 Toluene 300 432 559 X 110.4 1748 760 b- What is the mole fraction of benzene in the vapor phase (yb-?) at x -0.4 y 3. From the following vapor pressure data: a- Construct the Txy diagram using EXCEL at a total vapor pressure=1 atm, for the system benzene-toluene, assuming ideal solution behavior. Hint: use the following equations: PAP'S A. P= PA+ (PB-PAYA B. P= PB + (PA-PB)XA Temperature °C 80 92 100 Vapor pressure, mm Hg Benzene 760 1078 1344 Toluene 300 432 559 X 110.4 1748 760 b- What is the mole fraction of benzene in the vapor phase (yb-?) at x -0.4 y
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
What is the mole fraction of water vapor in air that is saturated with water at 25oC and 101.33 kPU? At 50oC and 10133 kPit?
-
What is the mole fraction of urea, CO(NH2)2, in a solution prepared by dissolving 4.8g of urea in 30.3g of methanol, CH3OH?
-
What is the mole fraction of toluene in a solution of 3.4 mol of benzene and 5.2 mol of toluene? A) 0.64 B) 0.54. C) 0.60. D) 0.24. E). 0.40
-
In Problem, p is the price per unit in dollars and q is the number of units. If the weekly demand function is p = 30 - q and the supply function before taxation is p = 6 + 2q, what tax per item will...
-
Consider Table 610 of the 2008 U.S. Statistical Abstract. (a) How many workers aged 20 or older were unemployed in the United States during 2006? How many of these were unemployed less than 5 weeks,...
-
explain the differences and similarities of the following terms: converse, inverse, and contrapositive statements. Provide an example to illustrate each of these terms. Reference the materials or...
-
Derive the following expression for determining \(\mathrm{K}_{\mathrm{y}} \mathrm{a}\) from the measurement of \(\mathrm{E}_{\mathrm{MV}}\) in a distillation column if the flow pattern is plug flow....
-
Alpine Company pays its employees time- and- a- half for hours worked in excess of 40 per week. The information available from time cards and employees individual earnings records for the pay period...
-
Good day, tutor! Could you please help me answer the following alphabetic sequences from the specific question in great detail? Preferably on paper, instead of using Excel. Please show your working...
-
How do you calculate the percentage of change in sales from 20X8 to 20X9? This will go in cell D7? Show the formula. Kretsmart Common-Size Income Statements For the Years Ended December 31, 20'X'9,...
-
3. (10 points) Find the length of the part of the curve 2 R(t) = t + + 1 t F between the origin and the point (1, 2/2, 1/3).
-
What is a post-mortem?
-
Of the information contained in a loan amortization schedule, what part is recorded in QuickBooks Accountant?
-
Lakeview Development Corporation was formed on January 2, 19X0, to develop a vacation-recreation area upon land purchased the same day by the corporation for \(\$ 100,000\). The corporation also...
-
Explain the difference between the Expenses tab and the Items tab in the Write Checks window.
-
The following situations involve Herb Standard, staff accountant with the regional CPA firm of Cash \& Green: 1. The bookkeeper of Ethical Manufacturing Company resigned two months ago and has not...
-
An experimenter is interested in the effect of a new drug on alertness. They gave half their participants a stimulant, while the other half received a placebo. Participants' alertness was measured...
-
Identify the source of funds within Micro Credit? How does this differ from traditional sources of financing? What internal and external governance mechanisms are in place in Micro Credit?
-
Using Appendix 4 and the following data, determine So for Fe(CO)5(g). Fe(s) + 5CO(g) Fe(CO)5(g) So = ? Fe(CO)5(l) Fe(CO)5(g) So = 107 J/ K Fe(s) + 5CO(g) Fe(CO)5(l) So = 677 J/ K
-
A standard galvanic cell is constructed so that the overall cell reaction is 2Al3+(aq) + 3M(s) 3M2+(aq) + 2Al(s) where M is an unknown metal. If Go = 411 kJ for the overall cell reaction, identify...
-
Equal moles of sulfur dioxide gas and oxygen gas are mixed in a flexible reaction vessel and then sparked to initiate the formation of gaseous sulfur trioxide. Assuming that the reaction goes to...
-
Which of the following is not an advantage of transferable pollution rights? a. They create incentives for polluters to develop cheaper ways to reduce pollution. b. They allow the greatest value of...
-
What is the relationship between externalities and property rights?
-
An advantage that corrective taxes and tradable emissions permits have over compliance standards is that the former a. works well even if pollution output cannot be accurately measured. b. results in...

Study smarter with the SolutionInn App



