4. (22 Points) The IR spectrum and proton NMR spectrum for a compound with molecular formula...
Fantastic news! We've Found the answer you've been seeking!
Question:
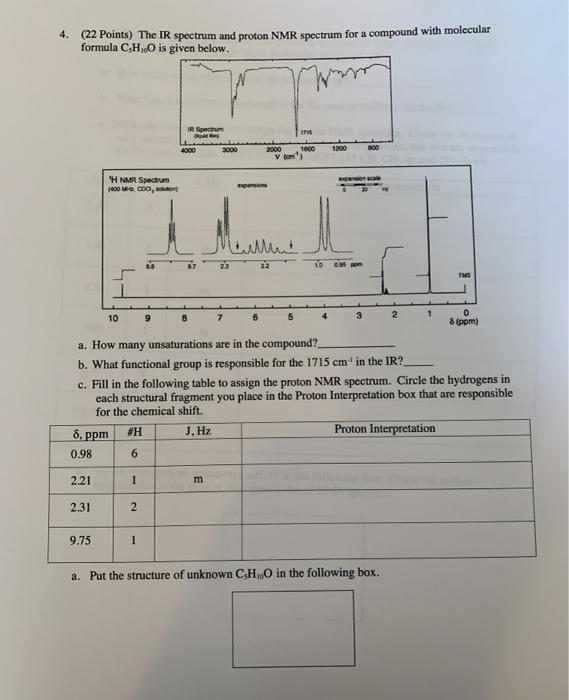
Transcribed Image Text:
4. (22 Points) The IR spectrum and proton NMR spectrum for a compound with molecular formula C,HO is given below. IR Spectrum this 4000 3000 2000 1600 1200 800 v (om) persion soale H NMR Spectrum (400 M CDO, on expansons 1o es om 10 6. 3. 8 (ppm) a. How many unsaturations are in the compound? b. What functional group is responsible for the 1715 cm in the IR? c. Fill in the following table to assign the proton NMR spectrum. Circle the hydrogens in each structural fragment you place in the Proton Interpretation box that are responsible for the chemical shift. #H J. Hz Proton Interpretation 8, ppm 0.98 6. 2.21 m 2.31 2. 9.75 a. Put the structure of unknown CH0 in the following box. 4. (22 Points) The IR spectrum and proton NMR spectrum for a compound with molecular formula C,HO is given below. IR Spectrum this 4000 3000 2000 1600 1200 800 v (om) persion soale H NMR Spectrum (400 M CDO, on expansons 1o es om 10 6. 3. 8 (ppm) a. How many unsaturations are in the compound? b. What functional group is responsible for the 1715 cm in the IR? c. Fill in the following table to assign the proton NMR spectrum. Circle the hydrogens in each structural fragment you place in the Proton Interpretation box that are responsible for the chemical shift. #H J. Hz Proton Interpretation 8, ppm 0.98 6. 2.21 m 2.31 2. 9.75 a. Put the structure of unknown CH0 in the following box.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The IR spectrum of a compound with molecular formula C5H8O was obtained in CCl4 and is shown in Figure 13.42. Identify the compound. Wavelenga qum) 15 16 14 3600 340) 3800 3300 3000 280K 2600 2400...
-
The 1 H NMR spectrum of a compound with molecular formula C 7 H 15 C l exhibits two signals with relative integration 2 : 3. Propose a structure for this compound.
-
The 1 H NMR spectrum of a compound with molecular formula C 7 H 14 O 3 exhibits only three signals, and all three signals appear above 2 ppm (downfield of 2 ppm) on the spectrum. Propose a structure...
-
I really would like to know the steps idk how to start this buttrying to look for the gross pays has been frustrating, pleasehelp! Each of the following workers is piece-rate workers at Golden Boats...
-
In one hour, Sue can produce 40 caps or 4 jackets and Tessa can produce 80 caps or 4 jackets. a. Calculate Sues opportunity cost of producing a cap. b. Calculate Tessas opportunity cost of producing...
-
The management of Wyatt Corporation is frustrated because its parent company, SRW Corporation, repeatedly rejects Wyatt's capital spending requests. These refusals led Wyatt's management to conclude...
-
The Cooper Furniture Company of Potomac, Maryland, assembles two types of chairs (Recliners and Rockers). Separate assembly lines are used for each type of chair. Classify each cost item (AI) as...
-
For the past several years, Abby Brown has operated a part-time consulting business from her home. As of June 1, 2012, Abby decided to move to rented quarters and to operate the business, which was...
-
Assume the numbers in the diagram correspond to an area, A, in the bounded region in which they are contained. 1 9 8 2 4 5 3 What numbers make up the intersection of the circle, triangle, and square?
-
The number of accidents on a road per day is recorded for 80 days, giving the following results. It is thought that the dataset models a Poisson distribution with a rate of 2.5 accidents per day....
-
Three different approaches to raising the standard of living of the poor are wage subsidies (whereby employers are subsidized by the government to pay higher wages to low wage employees), a...
-
Using the aggregate demand (AD), the short-run aggregate supply (SRAS), and the long-run aggregate supply (LRAS) curves, briefly explain how an open market purchase will affect the equilibrium price...
-
conduct a hazard ananlysis for each of the processes/activities for the created flow chart. the identified processes or CCP'S will now need to be assessed for hazards, as they are the areas of the...
-
A perfectly competitive firm faces a market price of $100 and has total cost of TC = 100 + 0.25q + 0.01q 2 . How much output (q) should this firm produce to maximize profits?
-
What type of metrics does Volkswagen of America use to measure the effectiveness of the IMC campaigns? Explain
-
Explain a decision-making model by heath and heath in their decisive book chapter 1? How could a decision-making technique aid in better decision-making? What resources, in terms of their usefulness...
-
The infamous case of the sociologist as Voveur An investigation that has almost achieved particular notoriety because of its ethics is Humphreys' ( 1 9 7 0 ) infamous study of homosexual encounters...
-
Find the intercepts and then graph the line. (a) 2x - 3y = 6 (b) 10 - 5x = 2y
-
How can 1,2-, 1,3-, and 1,4-dinitrobenzene be distinguished by a. 1H NMR spectroscopy? b. 13C NMR spectroscopy?
-
Show how each of the following compounds can be synthesized from benzene: a. p-chloroaniline b. m-chloroaniline c. p-nitrobenzoic acid d. m-nitrobenzoic acid e. m-bromopropylbenzene f....
-
Show how propyl propionate could be prepared, using allyl alcohol as the only source of carbon.
-
See Table 2.5 showing financial statement data and stock price data for Mydeco Corp. a. Compute Mydecos PE ratio each year from 2019 to 2023. In which year was it the highest? b. What was Mydecos...
-
See Table 2.5 showing financial statement data and stock price data for Mydeco Corp. a. Compute Mydecos ROE each year from 2019 to 2023. b. Compute Mydecos ROA each year from 2019 to 2023. c. Which...
-
In early 2018, United Airlines (UAL) had a market capitalization of \($25.14\) billion, debt of \($12.73\) billion, and cash of \($5.59\) billion. United also had annual revenues of \($38.72\)...

Study smarter with the SolutionInn App



