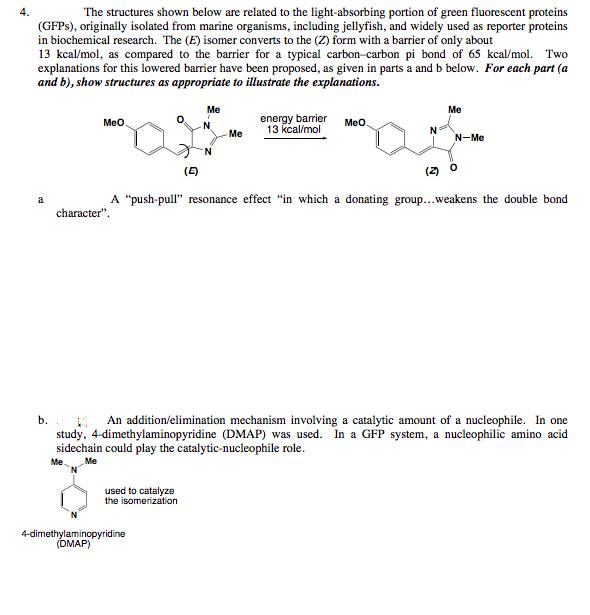
4. The structures shown below are related to the light-absorbing portion of green fluorescent proteins (GFPS),...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
4. The structures shown below are related to the light-absorbing portion of green fluorescent proteins (GFPS), originally isolated from marine organisms, including jellyfish, and widely used as reporter proteins in biochemical research. The (E) isomer converts to the (Z) form with a barrier of only about 13 kcal/mol, as compared to the barrier for a typical carbon-carbon pi bond of 65 kcal/mol. Two explanations for this lowered barrier have been proposed, as given in parts a and b below. For each part (a and b), show structures as appropriate to illustrate the explanations. Me Me energy barrier 13 kcal/mol Meo. N Meo Me N-Me (E) (2) O A "push-pull" resonance effect "in which a donating group...weakens the double bond a character". b. An addition/elimination mechanism involving a catalytic amount of a nucleophile. In one study, 4-dimethylaminopyridine (DMAP) was used. In a GFP system, a nucleophilic amino acid sidechain could play the catalytic-nucleophile role. Me Me N. used to catalyze the isomerization 4-dimethylaminopyridine (DMAP) 4. The structures shown below are related to the light-absorbing portion of green fluorescent proteins (GFPS), originally isolated from marine organisms, including jellyfish, and widely used as reporter proteins in biochemical research. The (E) isomer converts to the (Z) form with a barrier of only about 13 kcal/mol, as compared to the barrier for a typical carbon-carbon pi bond of 65 kcal/mol. Two explanations for this lowered barrier have been proposed, as given in parts a and b below. For each part (a and b), show structures as appropriate to illustrate the explanations. Me Me energy barrier 13 kcal/mol Meo. N Meo Me N-Me (E) (2) O A "push-pull" resonance effect "in which a donating group...weakens the double bond a character". b. An addition/elimination mechanism involving a catalytic amount of a nucleophile. In one study, 4-dimethylaminopyridine (DMAP) was used. In a GFP system, a nucleophilic amino acid sidechain could play the catalytic-nucleophile role. Me Me N. used to catalyze the isomerization 4-dimethylaminopyridine (DMAP)
Expert Answer:

Related Book For 

Organic Chemistry A Short Course
ISBN: 978-1111425562
13th edition
Authors: Harold Hart, Christopher M. Hadad, Leslie E. Craine, David J. Hart
Posted Date:
Students also viewed these chemistry questions
-
The costs listed below are related to a manufacturer of all-natural ice cream. In the space provided, indicate whether the cost should be classified as direct materials (DM), direct labor (DL), or...
-
2. The members of the structures shown below are made of plain concrete. The compressive strength of concrete is 4000 psi. Calculate the maximum P that can be carried by these structures. (Don't use...
-
A typical amino acid with one amino group and one carboxylic acid group, such as serine (Figure 24.18), can exist in water in several ionic forms. (a) Suggest the forms of theamino acid at low pH and...
-
The separation of operational responsibility from record keeping is meant to prevent different types of misstatements than the separation of the custody of assets from accounting. Explain the...
-
Using the data in the Excel file Freshman College Data, use ANOVA to determine whether significant differences exist in the mean retention rate for the different colleges over the 4 year period....
-
Suppose a dictionary named inventory exists. What does the following statement do? del inventory[654]
-
Provide an example of a known liability, an estimated liability, and a contingent liability.
-
In 2007, Charles Riegel and his wife sued a medical device manufacturer, Medtronic. Charles had a catheter produced by Medtronic placed in his coronary artery after he suffered a heart attack....
-
Cold One Brewing Company reported the following operating information for a recent year (in millions): Sales Cost of goods sold Gross profit Marketing, general, and admin. expenses Income from...
-
Wild West produces two types of cowboy hats. A Type 1 hat requires twice as much labor time as a Type 2. If all the available labor time is dedicated to Type 2 alone, the company can produce a total...
-
When considering all of the parts of the balance of payments for a nation, what item might you want to look at to determine if a nation has a trade surplus or deficit?
-
At some point for air flow in a duct, \(p=20 \mathrm{psia}, T=500^{\circ} \mathrm{R}\), and \(V=500 \mathrm{ft} / \mathrm{s}\). Can a normal shock occur at this point?
-
The landing speed of a winged aircraft such as the Space Shuttle is dependent on the air density. By what percent must the landing speed be increased on a day when the temperature is \(110^{\circ}...
-
Calculate the speed of sound in air, helium, and hydrogen. The temperature is \(70^{\circ} \mathrm{F}\).
-
A trout jumps, producing waves on the surface of a 0.8 -m-deep mountain stream. If it is observed that the waves do not travel upstream, what is the minimum velocity of the current?
-
What is the minimum water depth necessary for a 40 -ftwide stream to handle \(4000 \mathrm{ft}^{3} / \mathrm{s}\) if the flow is not supercritical?
-
The defendant's employees had been working in a manhole. They left the cover off the manhole when they went for lunch. They also left a paraffin lamp nearby. A young boy crawled down the manhole with...
-
Teasdale Inc. manufactures and sells commercial and residential security equipment. The comparative unclassified balance sheets for December 31, 2015 and 2014 are provided below. Selected missing...
-
Let us say that the (2R,3R) and (2S,3R) products in eq. 5.7 are formed in a 60:40 ratio. What products would be formed and in what ratio by adding HBr to pure (S)-3-chloro-1-butene? By adding HBr to...
-
D-Erythrose cannot exist in pyranose forms, but furanose cyclic forms are possible. Explain. Draw the structure for a-d-erythrofuranose.
-
Determine the order of reactivity for (CH3)2CHCH2Br, (CH3)3CBr, and in substitution reactions with a. Sodium cyanide. b. 50% aqueous acetone. CH CHCH2CH3 Br
-
The load resistor in the NMOS inverter in Figure \(16.3(\mathrm{a})\) is \(R_{D}=40 \mathrm{k} \Omega\). The circuit is biased at \(V_{D D}=3.3 \mathrm{~V}\). (a) Design the transistor...
-
The inverter circuit in Figure 16.3 (a) is biased at \(V_{D D}=3.3 \mathrm{~V}\). Assume the transistor conduction parameter is \(K_{n}=50 \mu \mathrm{A} / \mathrm{V}^{2}\). (a) Let \(R_{D}=100...
-
(a) Redesign the resistive load inverter in Figure 16.3 (a) so that the maximum power dissipation is \(0.25 \mathrm{~mW}\) with \(V_{D D}=3.3 \mathrm{~V}\) and \(v_{O}=0.15 \mathrm{~V}\) when the...

Study smarter with the SolutionInn App


