41.(6 pointa) In the following balanced reaction, magnesium metal (Mg) reacts with carbon dioxide gas (CO)...
Fantastic news! We've Found the answer you've been seeking!
Question:
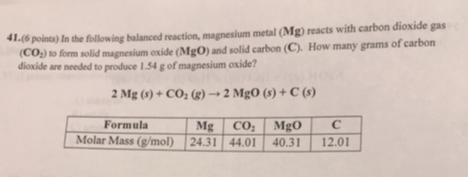
Transcribed Image Text:
41.(6 pointa) In the following balanced reaction, magnesium metal (Mg) reacts with carbon dioxide gas (CO) o form solid magnesium exide (MgO) and solid carbon (C). How many grams of carbon dioxide are needed to produce 1.54 g of magnesium oxide? 2 Mg (9) + CO, (2) 2 Mg0 (s) + C (s) Mg CO; Molar Mass (g/mol) 24.31 44.01 Formula MgO 40.31 12.01 41.(6 pointa) In the following balanced reaction, magnesium metal (Mg) reacts with carbon dioxide gas (CO) o form solid magnesium exide (MgO) and solid carbon (C). How many grams of carbon dioxide are needed to produce 1.54 g of magnesium oxide? 2 Mg (9) + CO, (2) 2 Mg0 (s) + C (s) Mg CO; Molar Mass (g/mol) 24.31 44.01 Formula MgO 40.31 12.01
Expert Answer:
Answer rating: 100% (QA)
An We have balanced egualion a ng so we woold need ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
How many grams of sulfur (S) are needed to react completely with 246 g of mercury (Hg) to form HgS?
-
1. How many grams of potassium hydroxy are needed to prepare 600 mL of 0.450 M solution? 2. What is the molarity of a solution prepared by dissolving 7.50 g of Mg(NO3), in enough water to form 25 mL...
-
How many grams of oxygen gas are required to produce 7.60 kJ of heat when hydrogen gas burns at constant pressure to produce gaseous water? 2H2(g) + O2(g) 2H2O(g); H = 484 kJ Liquid water has a heat...
-
In Exercises use the differential equation and the specified initial condition to find y. dy dx || 1 4 - x y(0) = -
-
Use the following information and deter mine the carrying amount of the property, plant, and equipment of Manotick Inc. on December 31, 2017, the company's year-end. (An accounting equation...
-
How do asymmetric information and behavioral economics influence the strategic decision-making process, ultimately shaping firms' ability to outmaneuver competitors and secure superior market...
-
Give three specialized subclasses for the Student class.
-
Shop Rite Services is ready to prepare its financial statements for the year ended December 31, 2012. The following information can be determined by analyzing the accounts: 1. On August 1, 2012, Shop...
-
A ball of mass 0,5 kg is projected vertically downwards from a height of 1,8 m with an initial velocity of 2 m.s1. The ball hits the ground and bounces two times. The position-time graph for the...
-
On October 1, 2020, Mary Graham organized a computer service company called Echo Systems. Echo is organized as a sole proprietorship and will provide consulting services, computer system...
-
Whirly Corporation s contribution format income statement for the most recent month is shown below: Total Per Unit Sales ( 7 , 4 0 0 units ) $ 2 3 6 , 8 0 0 $ 3 2 . 0 0 Variable expenses 1 3 3 , 2 0...
-
The assessment focuses on analysing the diversity policies and practices of the STARBUCKS The following information should be included in the report: Introduction to the business Identification of...
-
How can you address an employee who may not be keeping themselves up to standards? Pretend you need to have that conversation with an employee who has had a fellow employee report to you that they...
-
Analyse strategies for managing team leaders Strategies are important for managers in order to effectively lead a team. Examples of effective strategies to manage the team Delegation- What is...
-
Develop approaches to respond to the challenges of managing and leading multiple and remote teams Challenges include Communication ( methods , type , times , frequency, availability, interpretation...
-
David y Hope Smith planean presentar una declaracin de impuestos. Estn casados y tienen dos hijos, Katelyn y Cory. Todos viven en 134 Crystal Lake Road, New Hope, PA 18938. La familia Smith tena...
-
Abraham Maslow identified five human needs. The author of your textbook added a sixth need. Identify this sixth need and explain why this need is important to life in a diverse society ?
-
What is beacon marketing? What are digital wallets?
-
An antiseptic solution contains hydrogen peroxide, H2O2, in water. The solution is 0.600 m H2O2. What is the mole fraction of hydrogen peroxide?
-
Rate laws are not restricted to chemical systems; they are used to help describe many everyday events. For example, a rate law for tree growth might look something like this: Rate of growth = (soil...
-
The following data were collected for the reaction A(g) + B(g) Products. a. Determine the rate law for this reaction. b. Calculate the rate constant. c. Calculate the rate when [A] = 0.200 M and [B]...
-
A light ray travels from medium l to medium 3 as shown. For these media, A. nz > n 13 B. n3 = n nz C. n3
-
A Jens produces a sharply focused, inverted image on a screen. What will you see on the screen if the lens is removed? A. The image will be inverted and blurry. B. The image will be upright and...
-
An object and lens are positioned to form a well-focused, inverted image on a viewing screen. Then a piece of cardboard is lowered just in front of the lens to cover the top half of the lens. Using...

Study smarter with the SolutionInn App


