6 ch 1 12 4 15 Calculate the molar mass of each element or compound below....
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
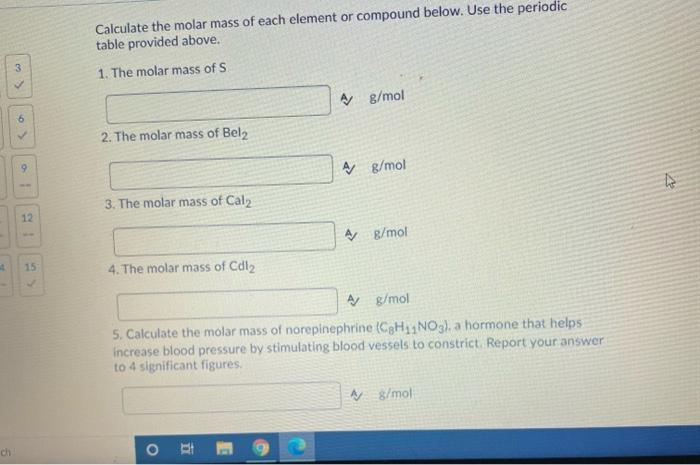
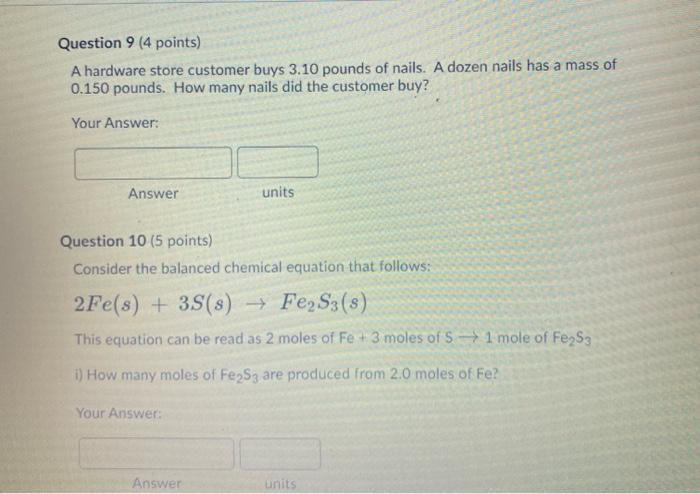
6 ch 1 12 4 15 Calculate the molar mass of each element or compound below. Use the periodic table provided above. 1. The molar mass of S 2. The molar mass of Bel2 3. The molar mass of Cal2 4. The molar mass of Cdl₂ O A g/mol HI A g/mol A g/mol 5. Calculate the molar mass of norepinephrine (C8H₁1NO3), a hormone that helps increase blood pressure by stimulating blood vessels to constrict. Report your answer to 4 significant figures. A g/mol A g/mol V 1: 3 9 11 12 14 15 Question 8 (5 points) Look at the units of molar mass and describe what kind of conversion factor it is and what it helps you do? fº Y Paragraph V BIU *** 9 Fax / Question 9 (4 points) A hardware store customer buys 3.10 pounds of nails. A dozen nails has a mass of 0.150 pounds. How many nails did the customer buy? Your Answer: Answer Question 10 (5 points) Consider the balanced chemical equation that follows: 2Fe(s) + 3S(s) → Fe₂S3(s) This equation can be read as 2 moles of Fe + 3 moles of S1 mole of Fe₂S3 i) How many moles of Fe2S3 are produced from 2.0 moles of Fe? Your Answer: units Answer units 3 6 19 1 12 15 Question 11 (5 points) Consider the balanced chemical equation that follows: 2Fe(s) + 3S(s) → Fe₂S3(s) This equation can be read as 2 moles of Fe + 3 moles of S→ 1 mole of Fe2S3 ii) How many moles of Fe2S3 are produced from 29.0 moles of S? Your Answer: Answer units 2 3 ✓ ✓ 55 6 ✓ 8 9 11 12 14 15 Question 12 (5 points) Consider the balanced chemical equation that follows: 2Fe(s) + 3S(s) → Fe2S3 (8) This equation can be read as 2 moles of Fe + 3 moles of 5→ 1 mole of Fe2S3 iii) How many moles of S react with 7.0 moles of Fe? Your Answer: Answer units 5 ✓ ✓ 8 11 6> 14 9. 12 15 Question 14 (4 points) Iron (III) oxide reacts with carbon to give iron and carbon monoxide: Fe2O3(s) + 3C(s)→2Fe(s) + 3CO(g) How many grams of C are required to react with 32.7 g of Fe₂O3 Your Answer: Answer units 6 ch 1 12 4 15 Calculate the molar mass of each element or compound below. Use the periodic table provided above. 1. The molar mass of S 2. The molar mass of Bel2 3. The molar mass of Cal2 4. The molar mass of Cdl₂ O A g/mol HI A g/mol A g/mol 5. Calculate the molar mass of norepinephrine (C8H₁1NO3), a hormone that helps increase blood pressure by stimulating blood vessels to constrict. Report your answer to 4 significant figures. A g/mol A g/mol V 1: 3 9 11 12 14 15 Question 8 (5 points) Look at the units of molar mass and describe what kind of conversion factor it is and what it helps you do? fº Y Paragraph V BIU *** 9 Fax / Question 9 (4 points) A hardware store customer buys 3.10 pounds of nails. A dozen nails has a mass of 0.150 pounds. How many nails did the customer buy? Your Answer: Answer Question 10 (5 points) Consider the balanced chemical equation that follows: 2Fe(s) + 3S(s) → Fe₂S3(s) This equation can be read as 2 moles of Fe + 3 moles of S1 mole of Fe₂S3 i) How many moles of Fe2S3 are produced from 2.0 moles of Fe? Your Answer: units Answer units 3 6 19 1 12 15 Question 11 (5 points) Consider the balanced chemical equation that follows: 2Fe(s) + 3S(s) → Fe₂S3(s) This equation can be read as 2 moles of Fe + 3 moles of S→ 1 mole of Fe2S3 ii) How many moles of Fe2S3 are produced from 29.0 moles of S? Your Answer: Answer units 2 3 ✓ ✓ 55 6 ✓ 8 9 11 12 14 15 Question 12 (5 points) Consider the balanced chemical equation that follows: 2Fe(s) + 3S(s) → Fe2S3 (8) This equation can be read as 2 moles of Fe + 3 moles of 5→ 1 mole of Fe2S3 iii) How many moles of S react with 7.0 moles of Fe? Your Answer: Answer units 5 ✓ ✓ 8 11 6> 14 9. 12 15 Question 14 (4 points) Iron (III) oxide reacts with carbon to give iron and carbon monoxide: Fe2O3(s) + 3C(s)→2Fe(s) + 3CO(g) How many grams of C are required to react with 32.7 g of Fe₂O3 Your Answer: Answer units
Expert Answer:
Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
Calculate the molar mass of the following substances: (a) Li2 CO3, (b) CS2, (c) CHCl3 (chloroform), (d) C6H8O6 (ascorbic acid, or vitamin C), (e) KNO3, (f) Mg3N2.
-
What is the molar mass of AgNO3?
-
What is the molar mass of a gas whose density is 168 gL at STP.
-
Maggie, Bihun, and Mee decided to go into the landscaping business. The three orally declared that they would be partners in PPL Landscapers, sharing profits and losses equally. Maggie, Bihun, Mee...
-
Define the term leverage as it applies to accounting.
-
How do ethical considerations surrounding corporate responsibility, stakeholder stewardship, and societal welfare intersect with the exigencies of bankruptcy law and financial restructuring...
-
Explain the importance of a written contract.
-
The December 31, 2012, statement of financial position for the Blood Donors of America Foundation is presented below. Statement of Financial Position December 31, 2012 Assets...
-
Your beginning salary is $68,000. You deposit 8% each year in a savings account that earns 3% interest. Your salary increases by 5% per year and inflation is 2.5% per year. What value does your...
-
Find and classify the stationary values of the function f(x, y) = sin x cos y + 2 cos x sin y in the range 0xm, x y 0syn. fxy 2 fxxfyy-fxy Type f(x,y)
-
A hail storm caused nearly golf-ball sized hail to fall on a house. The hailstones were nearly spherical, with a diameter of roughly 3.7 cm and a weight of 24 g. a. Draw a free body diagram of the...
-
Samuel just moved to Radford and plans to live here for 5 years. He just found a "perfect" house to live in with his family. The current owner is willing to sell or rent the house to Samuel. The...
-
ABC is now considering changing the debt ratio and moving it to the new debt/assets ratio as indicated below, and replacing all preferred stocks with debt. The money raised would be used to...
-
how to test for equal population variance. In the same setting, perform a two- sided test on Ho: -20%=0. Derive the test statistic, define the critical value you need, and specify the rejection...
-
How would you solve for this? You have been asked to conduct a valuation of 4 companies using their Free Cash Flow to Firm (FCFF). Assuming that all companies experience supernormal growth per table...
-
Consider an oligopoly in which firms choose quantities. The inverse market demand curve is given by P = a - b (q + 92), where q is the quantity produced by Firm 1, and q2 is the quantity produced by...
-
Issuance of bond with 12 percent coupon. The floatation cost is 5 percent of the RM1,245 market value. The maturity period is 10 years and the tax rate of the company is 35 percent. Compute the cost...
-
Test whether the 5-year survival rate for breast cancer is significantly different between African American and Caucasian women who are younger than 50 years of age and have localized disease....
-
In 2012, Michael has net short-term capital losses of $2,000, a net long-term capital loss of $45,000, and other ordinary taxable income of $45,000. a. Calculate the amount of Michael's deduction for...
-
Patty Bayan is a single taxpayer living at 543 Space Drive, Houston, TX 77099. Her Social Security number is 466-33-1234. For 2012, Patty has no dependents, and her W-2 from her job at a local...
-
During 2012, Tom sold Sears stock for $10,000. The stock was purchased 4 years ago for $13,000. Tom also sold Ford Motor Company bonds for $35,000. The bonds were purchased 2 months ago for $30,000....
-
Reconsider the data of Problem 31. Data from Problem 31 The following three investment opportunities are available. The returns for each investment for each year vary, but the first cost of each is...
-
The following three investment opportunities are available. The returns for each investment for each year vary, but the first cost of each is $20,000. Based on a future worth analysis, which...
-
Explain the concept of a control variable and the assumption necessary for a control variable to be effective.

Study smarter with the SolutionInn App



