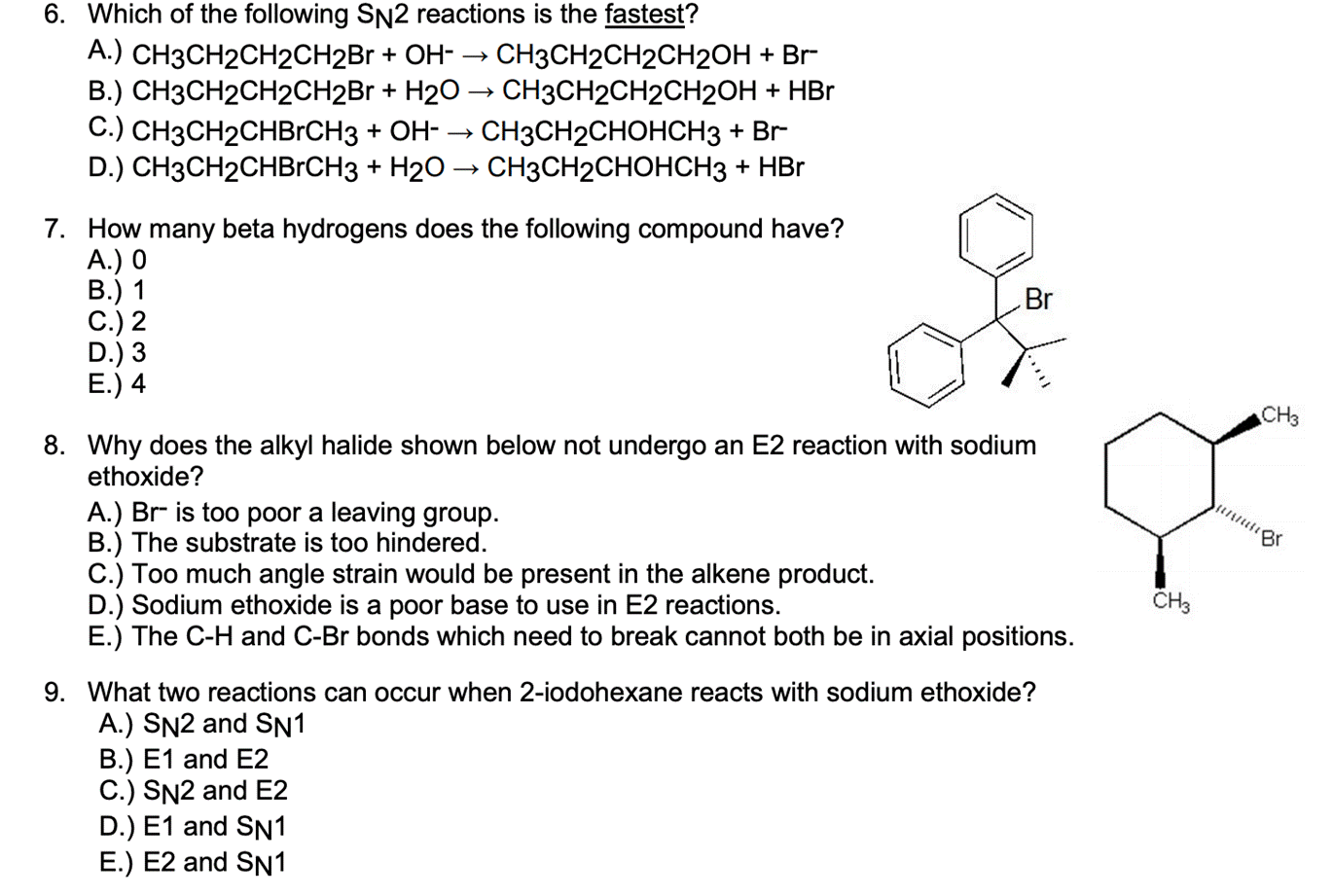
6. Which of the following SN2 reactions is the fastest? A.) CH3CH2CH2CH2BR + OH- B.) CH3CH2CH2CH2B...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
6. Which of the following SN2 reactions is the fastest? A.) CH3CH2CH2CH2BR + OH- B.) CH3CH2CH2CH2B + H20 → CH3CH2CH2CH2OH + HBr C.) CH3CH2CHBRCH3 + OH- → D.) CH3CH2CHBRCH3 + H20 → CH3CH2CHOHCH3 + HBr CH3CH2CH2CH2OH + Br CH3CH2CHOHCH3 + Br 7. How many beta hydrogens does the following compound have? A.) 0 В.) 1 C.) 2 D.) 3 E.) 4 Br CH3 8. Why does the alkyl halide shown below not undergo an E2 reaction with sodium ethoxide? A.) Br is too poor a leaving group. B.) The substrate is too hindered. C.) Too much angle strain would be present in the alkene product. D.) Sodium ethoxide is a poor base to use in E2 reactions. E.) The C-H and C-Br bonds which need to break cannot both be in axial positions. 9. What two reactions can occur when 2-iodohexane reacts with sodium ethoxide? A.) SN2 and SN1 B.) E1 and E2 C.) SN2 and E2 D.) E1 and SN1 E.) E2 and SN1 6. Which of the following SN2 reactions is the fastest? A.) CH3CH2CH2CH2BR + OH- B.) CH3CH2CH2CH2B + H20 → CH3CH2CH2CH2OH + HBr C.) CH3CH2CHBRCH3 + OH- → D.) CH3CH2CHBRCH3 + H20 → CH3CH2CHOHCH3 + HBr CH3CH2CH2CH2OH + Br CH3CH2CHOHCH3 + Br 7. How many beta hydrogens does the following compound have? A.) 0 В.) 1 C.) 2 D.) 3 E.) 4 Br CH3 8. Why does the alkyl halide shown below not undergo an E2 reaction with sodium ethoxide? A.) Br is too poor a leaving group. B.) The substrate is too hindered. C.) Too much angle strain would be present in the alkene product. D.) Sodium ethoxide is a poor base to use in E2 reactions. E.) The C-H and C-Br bonds which need to break cannot both be in axial positions. 9. What two reactions can occur when 2-iodohexane reacts with sodium ethoxide? A.) SN2 and SN1 B.) E1 and E2 C.) SN2 and E2 D.) E1 and SN1 E.) E2 and SN1
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
How will the rate of each of the following SN2 reactions change if the polarity of a protic polar solvent is increased? a. b. c. CH,CH2CH2CH2BrHOTCH CH2CH2CH2OHBr CH3 CH,CH31 + NH3 CH3CH,NH, -
-
How many signals would be present in the 1H NMR spectrum of each of the cyclopropane derivatives shown below? Consider carefully the geometric environments around each hydrogen. (a) (b) (c) (d) (e)...
-
What types of marketing opportunities would be present in BRIC countries that would not be available in least developed economies? Would the number of bottom-of-the-pyramid consumers in either...
-
An individual has capital losses brought forward from previous years amounting to 4,800. Compute the individual's taxable gains for 2020-21 if total gains and losses for the year are as follows:...
-
The U. S. Tobacco Settlement between the major tobacco companies and 46 states caused the price of cigarettes to jump 45 (21%) in November 1998. Levy and Meara (2005) found only a 2.65% drop in...
-
On January 1 of this year, Kona Corporation sold bonds with a face value of $1,440,000 and a coupon rate of 10 percent. The bonds mature in four years and pay interest semiannually every June 30 and...
-
The equilibrium criterion of a chemical reaction can be expressed as (a) \((d G)_{T, P}=1\) (b) \((d G)_{T, P}=-1\) (c) \((d G)_{T, P}=0\) (d) \((d G)_{T, P}=\infty\).
-
The following transactions pertain to the operations of Ewing Company for 2016: 1. Acquired $30,000 cash from the issue of common stock. 2. Provided $65,000 of services on account. 3. Paid $22,000...
-
4. Krawl Company uses a job order cost system. The following data summarizes the operations related to production for June 20X1, the first month of operations. a. Materials purchased on account for...
-
Will Googles pay strategy work forever? Consider the evolution of Microsofts pay strategy as its growth slackened. Should Google prepare for a similar future? If so, when and what sort of actions...
-
Question 7 (4 points) Please use Software Expert (SWE) Database. List the first and last name of every consultant who has ever worked on a project with consultant Mark Myers. Include Mark Myers in...
-
A production line is to be designed to assemble a product. The assembly of this product requires three tasks, one of which requires 0.3 minutes to complete, one of which requires 1.4 minutes to...
-
(a) Distinguish between 'non-controllable' and 'controllable' risk. (b) Explain briefly the components of non-controllable and controllable risk.
-
Why would sustainability alone not necessarily guarantee the survival of an organization?
-
The North Atlantic Consortium of Nephrology Associations (NACNA) is made up of two professional societies, the North American Nephrologist Society (NANS) and the European Council on Nephrology (ECN)....
-
Does a service operation often require higher or lower capital investment at start-up, when compared to goods production?
-
Using the experimentally obtained IR spectra, tabulate the IR frequencies and intensities for both cis and trans isomers of copper(II) glycine complexes. Make necessary functional group assignments...
-
A bar of a steel alloy that exhibits the stress-strain behavior shown in Figure 6.22 is subjected to a tensile load; the specimen is 375 mm (14.8 in.) long and has a square cross section 5.5 mm (0.22...
-
Draw the enol tautomers for each of the following compounds. For those compounds that have more than one enol tautomer, indicate which is more stable. a. b. c. d. e. f. CH3CH2CCH,CH3 CCH3 CH CH...
-
Show how the following compounds could be prepared, starting with 3 cyanocyclohexanone a. b. CH2CH2CCH
-
Show the aldol addition product that would be formed from each of the following compounds: a. b. c. d. CHCHCH-CHCH - - CH CHCH2CH CH CH3 ,
-
What is the difference between old classical and new classical economics?
-
What is the difference between traditional Keynesian and new Keynesian economics?
-
Why does monetary policy operate with a long and variable lag? Give an example to illustrate your explanation.

Study smarter with the SolutionInn App



