6:13 2 of 5 A < The pH of a vinegar solution is 5.72. What is...
Fantastic news! We've Found the answer you've been seeking!
Question:
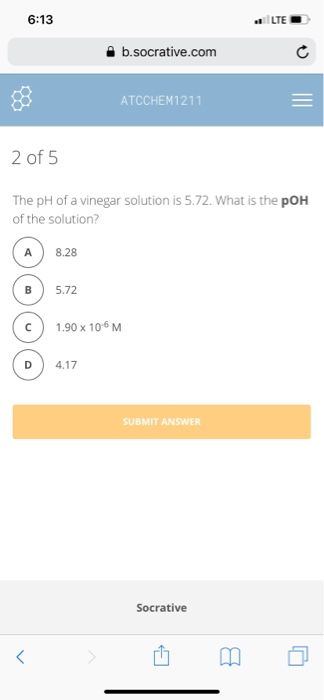
Transcribed Image Text:
6:13 2 of 5 A < The pH of a vinegar solution is 5.72. What is the pOH of the solution? 8.28 5.72 b.socrative.com ATCCHEM1211 4.17 1.90 x 10-6 M SUBMIT ANSWER LTE Socrative 6:13 2 of 5 A < The pH of a vinegar solution is 5.72. What is the pOH of the solution? 8.28 5.72 b.socrative.com ATCCHEM1211 4.17 1.90 x 10-6 M SUBMIT ANSWER LTE Socrative
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A solution of vinegar is 0.763 M acetic acid, HC2H3O2. The density of the vinegar is 1.004 g/mL. What is the molal concentration of acetic acid?
-
The pH of a white vinegar solution is 2.45. This vinegar is an aqueous solution of acetic acid with a density of 1.09 g/mL. What is the mass percentage of acetic acid in the solution?
-
A 4 m x 5 m x 6 m room contains 120 kg of air. Determine (a) Density. (b) Specific volume. (c) Mole (d) Specific molar volume of air. Assume molar mass of air to be 29 kg/kmol.
-
Let Z[] be the following set of rational numbers { a Z, k N} (and recall that N = {0, 1, 2,...} in this class). Let the addition and multiplication for Z[] be the usual addition and multiplication...
-
Deacon Co. reported annual net sales for 2010 and 2011 of $565,000 and $647,000, respectively. Its yearend balances of accounts receivable follow: December 31, 2010, $51,000; and December 31, 2011,...
-
How has Amazon Prime demonstrated the truth of the statement that demand management and customer service are closely linked? (Remember that order fulfillment is a key determinant of how satisfied...
-
The stockholders' equity of Lakewood Occupational Therapy, Inc., on December 31, 2009, follows. On April 30, 2010, the market price of Lakewood's common stock was $14 per share and the company...
-
Campbell Corporation reports regular taxable income of $210,000 in the current year. Campbell takes into account the following facts when calculating the $210,000 amount. Campbell deducts $100,000...
-
White Financial Services Corporation is engaged in mortgage origination and investment servicing activities with annual revenues of more than $90 million. Roberta White, CEO, recently has discovered...
-
Background: SO MUCH CANDY DATA, SERIOUSLYCandy hierarchy data for 2017 Boing Boing Halloween candy hierarchy. This is survey data over the span of 4-years. The data is split into 4 separate files....
-
public class MySqrtMethod ( public static void main(String[] args) { } // read in the command-line argument double x Double.parseDouble (args[0]); /* Newton's method to calculate square root of a...
-
Please discuss all three (3) statements utilizing your textbook, all other materials provided. Provide 'complete answers' to the questions - indicating your knowledge of the subject matter supported...
-
The grand management paradox that systems thinkers face is a result of the tension that exists between the forces of static equilibrium or the resistance to change and dynamic equilibrium or being...
-
Will your company warranty your products? Why or why not? What, if any, express warranties will your company offer? To what, if any, implied warranties will your company be subject? If you are not...
-
The structure in the figure below is made up of a cantilever beam AB (E, I, A) and two identical rods BC and CD (E2, A). Let A, be large compared with A and L, be large compared with the beam depth....
-
What is the significance of log auditing and monitoring in operating system security? How do advanced persistent threats (APTs) attempt to evade detection through log tampering or obfuscation?
-
Question: Propose a sustainable strategic asset allocation for the entire direct investment portfolio of OHPIB by producing a report. The case is intentionally left open along many dimensions and you...
-
D Which of the following is considered part of the Controlling activity of managerial accounting? O Choosing to purchase raw materials from one supplier versus another O Choosing the allocation base...
-
Obtain the moles of substance in the following. a. 2.86 g C b. 7.05 g Cl2 c. 76 g C4H10 d. 26.2 g Al2(CO3)3
-
An antiseptic solution contains hydrogen peroxide, H2O2, in water. The solution is 0.600 m H2O2. What is the mole fraction of hydrogen peroxide?
-
What is meant by the precision of a measurement? How is it indicated?
-
Derive the equation of motion and natural frequency for a mass \(m\) on the string that is under constant tension \(T\) as shown in Figure 2.44. Assume small displacements and that \(m\) is much...
-
Continuing Problem 13, the string is stretched to the position shown in Figure 2.45. Calculate the natural frequency of the system using the following parameter values: \(m g=2 \mathrm{lb}, T=50...
-
Derive the equation of motion for a uniform stiff rod restrained from vertical motion by a torsional spring of stiffness \(K\) and two translational springs each of stiffness \(k\), as shown in...

Study smarter with the SolutionInn App



