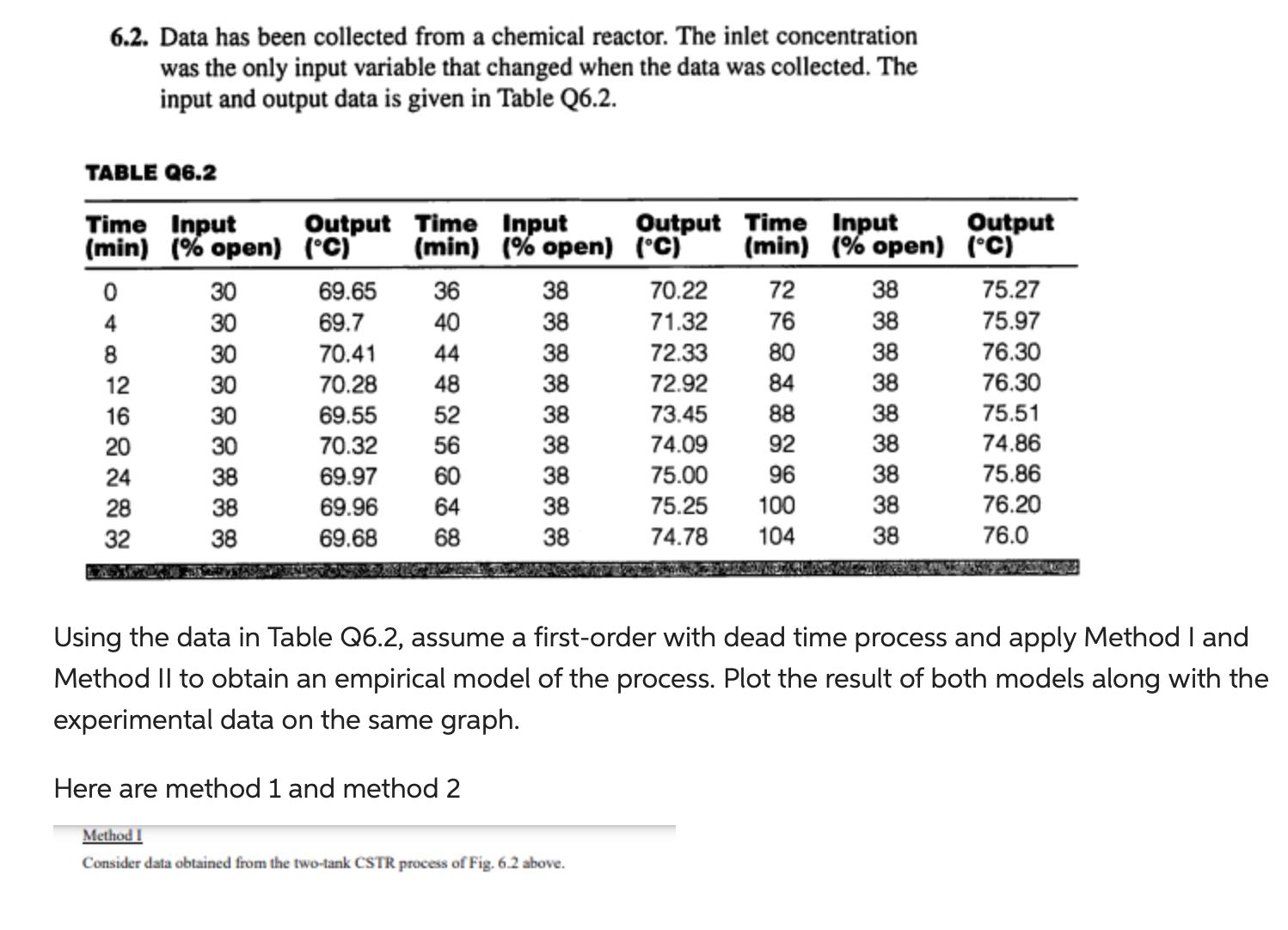
6.2. Data has been collected from a chemical reactor. The inlet concentration was the only input...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
6.2. Data has been collected from a chemical reactor. The inlet concentration was the only input variable that changed when the data was collected. The input and output data is given in Table Q6.2. TABLE Q6.2 Time Input (min) (% open) 0 4 8 12 16 20 24 28 32 30 30 30 30 30 30 38 38 38 Output Time Input (C) 69.65 69.7 70.41 70.28 69.55 70.32 69.97 69.96 69.68 (min) (% open) 36 40 44 48 52 56 60 64 68 38 38 38 38 38 38 Here are method 1 and method 2 38 38 38 Output Time Input (C) (min) (% open) Method I Consider data obtained from the two-tank CSTR process of Fig. 6.2 above. 70.22 71.32 72.33 72.92 73.45 74.09 75.00 75.25 100 74.78 104 72 76 80 84 88 92 96 38 38 38 38 38 38 38 38 38 Output (C) Using the data in Table Q6.2, assume a first-order with dead time process and apply Method I and Method II to obtain an empirical model of the process. Plot the result of both models along with the experimental data on the same graph. 75.27 75.97 76.30 76.30 75.51 74.86 75.86 76.20 76.0 6.2. Data has been collected from a chemical reactor. The inlet concentration was the only input variable that changed when the data was collected. The input and output data is given in Table Q6.2. TABLE Q6.2 Time Input (min) (% open) 0 4 8 12 16 20 24 28 32 30 30 30 30 30 30 38 38 38 Output Time Input (C) 69.65 69.7 70.41 70.28 69.55 70.32 69.97 69.96 69.68 (min) (% open) 36 40 44 48 52 56 60 64 68 38 38 38 38 38 38 Here are method 1 and method 2 38 38 38 Output Time Input (C) (min) (% open) Method I Consider data obtained from the two-tank CSTR process of Fig. 6.2 above. 70.22 71.32 72.33 72.92 73.45 74.09 75.00 75.25 100 74.78 104 72 76 80 84 88 92 96 38 38 38 38 38 38 38 38 38 Output (C) Using the data in Table Q6.2, assume a first-order with dead time process and apply Method I and Method II to obtain an empirical model of the process. Plot the result of both models along with the experimental data on the same graph. 75.27 75.97 76.30 76.30 75.51 74.86 75.86 76.20 76.0
Expert Answer:
Answer rating: 100% (QA)
To obtain an empirical model of the process using a firstorder with dead time model we will apply Me... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
A researcher wanted to find out if there was difference between older movie goers and younger movie goers with respect to their estimates of a successful actors income. The researcher first...
-
My question is how to write the code in WareHouse.java This is WareHouse.java package warehouse; /* * * This class implements a warehouse on a Hash Table like structure, * where each entry of the...
-
Adel runs a construction equipment rental company, renting out loaders, generators, lifts and similar equipment on short term rentals. In the past, Adel has purchased the equipment he then rents out,...
-
Vector A points in the negative y direction and has a magnitude of 5 units. Vector B has twice the magnitude and points in the positive x direction. Find the direction and magnitude of (a) A(vector)...
-
From inception of operations to December 31, 2020, Fortner Corporation provided for uncollectible accounts receivable under the allowance method. The provisions are recorded, based on analyses of...
-
The results of longitudinal, transverse, and \(45^{\circ}\) off-axis tensile tests on samples from an orthotropic lamina are shown in Figure 10.35. Based on these results, find numerical values for...
-
1. What are some things Colin could have done in or after the meeting when Henri verbally attacked him? 2. Is there anything more Raouf could have done during or after his meeting with Colin to...
-
A regional electrical distributor currently has 1,000 customers who buy an average of $5,000 per year, generating a 50% margin. From experience, the company knows that 20% of its customers will not...
-
Euro Corporation, a U.S. corporation, operates through a branch in Germany. During 2022, the branch reported taxable income of $1,000,000 and paid German income taxes of $300,000. In addition, Euro...
-
Find an equation of the line that satisfies the conditions. Write the equation in standard form. 5 Through (2, 4); m = -5
-
The following diagram shows the Gint coefficient and income share of the B40. M40 and T20 groups in Malaysia between the years 1970 and 2019. Income Share (%) 70.0 60.0 55.7 50.0 40.0 30.0 20.0 10.0...
-
Suppose you serve demand in a set of six cities with a single product, where each city has weekly demand for the product that is normally distributed with mean 250 units and standard deviation 85....
-
On a typical day, the Stonebriar mall sees customers arriving at an average rate of 225 per hour. Each customer stays in the mall for an average of 135 minutes. 1. What is the average number of...
-
Phone Corporation owns a 90% interest in Style Company, acquired several years ago at a cost equal to book value and fair value. Style sells merchandise to Phone for the first time in 2023, and some...
-
Consider the following fragment of the Data Flow Graph (DFG) of the differential equation solver used in the course slides, HLS-scheduling; we are only concerned with output ul of the DFG. You are...
-
Youve just been appointed senior mortgage loan officer at the Wawa (Ontario) brach of the Royal Bank. A Ms. K. Wynne, who is a potential mortgagor, comes to you seeking a $750, 000 fixed-rate...
-
From a medical tourist perspective, compare Shouldice with the traditional hospital in terms of the key factors of competition. Using Table 15-3, why would Shouldice attract patients from outside the...
-
Derive Equation. d 2 yd 2 n 2 yn=0 by 2dy/d, rearrange to get dd(dyd) 2 =n 2 yn 2 dyd and solve using the boundary conditions dy/d = 0 at = 0.
-
What if you were asked for all the things that could go wrong in the operation of a slurry reactor as described in the Professional Reference Shelf for Chapter 15? What would you say?
-
Use the data in Problem P11-4A for the following reaction. The elementary, irreversible, organic liquid-phase reaction A + B C is carried out in a flow reactor. An equal molar feed in A and B enters...
-
Describe what sexual harassment is and what forms it can take.
-
Explain public policies that protect employees from unlawful discharge.
-
Discuss under what conditions unemployment compensation can be denied to a claimant.

Study smarter with the SolutionInn App


