7) Br heat + CH;CO,H (solvent acetic acid) 7) Br heat + CH;CO,H (solvent acetic acid)...
Fantastic news! We've Found the answer you've been seeking!
Question:
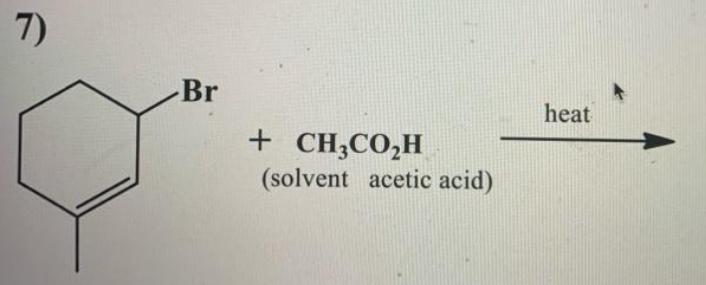
Transcribed Image Text:
7) Br heat + CH;CO,H (solvent acetic acid) 7) Br heat + CH;CO,H (solvent acetic acid)
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Acetic anhydride is formed from acetic acid in a condensation reaction that involves the removal of a molecule of water from between two acetic acid molecules. Write the chemical equation for this...
-
Acetic acid was evaporated in container of volume 21.45 cm3 at 437 K and at an external pressure of 101.9 kPa, and the container was then sealed. The mass of acid present in the sealed container was...
-
Acetic acid can be manufactured by combining methanol with carbon monoxide, an example of a carbonylation reaction: (a) Calculate the equilibrium constant for the reaction at 25°C. (b)...
-
A. Discuss the following conceptual model/framework of Orem's Self-Care Theory CONDITIONING FACTORS (12 MARKS) Orem's Self-Care Theory Conceptual Framework SELF-CARE AGENCY SELF-CARE DEFICIT NURSING...
-
1) Why has eBay succeeded as an online auction marketplace while so many others have failed? 2) Evaluate eBay's fee structure. Is it optimal or could it be improved? Why? How? 3) What's next for...
-
You purchase 10 call option contracts with a strike price of $75 and a premium of $4.05. If the stock price at expiration is $83.61, what is your dollar profit? What if the stock price is $69.56?
-
Bert C. Roberts Jr. was chairman of WorldComs board of directors. Immediately before that, he had been chairman of MCI, which WorldCom acquired on September 14, 1998, in a transaction valued at...
-
Clydesdale Corporation has a cumulative temporary difference related to depreciation of $580,000 at December 31, 2010. This difference will reverse as follows: 2011, $42,000; 2012, $244,000; and...
-
1. Write an integral using cylindrical coordinates that represents the volume between the surfaces z=x+ y and z = 6-x - y.
-
Assignment #1_ CO# S2 CHAPTER 9: LEADERSHIP AND CHANGE Flow Chart: Search the internet and obtain a "leadership" written procedure/approach/plan. Copy then paste it on the Excel embedded table below....
-
From the excess return formula, calculate the breakeven point knowing the following information: - Spot (or cash) exchange rate = $1.61/. -Investment of $10000 for one year. -U.S. interest = 7.90%...
-
When payment is delayed on a foreign transaction a seller may receive less revenue than expected at time of payment due to the risk of
-
Accepting only cashless payment forms, such as credit cards or mobile app payments, is an internal control that mitigates the risk of cash being stolen by a cashier in charge of a cash register....
-
Mrs. Tolentino is reviewing the clients chart to determine if the degree of nursing care was satisfactorily rendered according to prescribed standards which falls under Nursing audit C. Prospective...
-
provides a breakdown of the components of the total interest expense on its Convertible Notes. How did the existence of $3,001 of Amortization of discount on its convertible "2026 Notes" during...
-
a. One tool of corporate governance is monitoring management. b. One tool of corporate governance is the choice of how much dividends to pay. c. Corporate governance is better when Directors are also...
-
Johnson Ltd. determined that the net present value of an investment in technological improvements at its plant in France would be 12,000,000 if pending litigation was resolved in the company's...
-
Discuss the concept of the looking-glass self. how do you think others perceive you? do you think most people perceive you correctly?
-
Boric acid, B(OH)3, is used as a mild antiseptic. What is the pH of a 0.021 M aqueous solution of boric acid? What is the degree of ionization of boric acid in this solution? The hydronium ion arises...
-
What is the relationship between bond order and bond length? Use an example to illustrate it.
-
Calcium oxide (quicklime) reacts with water to produce calcium hydroxide (slaked lime). CaO(s) + H2O(l ) Ca(OH)2(s); H = 65.2 kJ The heat released by this reaction is sufficient to ignite paper. How...
-
Martin Galloway, the sole proprietor of a consulting business, has gross receipts of $45,000 in 2017. His address is: 1223 Fairfield Street, Westfield, New Jersey and his SSN is 158-68-7799. Expenses...
-
Probest Corporation (a calendar-year corporation) purchased and placed the following assets in service during the year All assets are used 100 percent for business. $100,000 of the cost of the...
-
In May 2016, Jose purchased a used automobile for $12,000 and used it 75 percent for business. No Section 179 election was made for this asset. In 2017, Joses business use of the automobile decreased...

Study smarter with the SolutionInn App



