7. Techniques of protein purification a. Why is SDS omitted when proteins need to undergo isoelectric...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
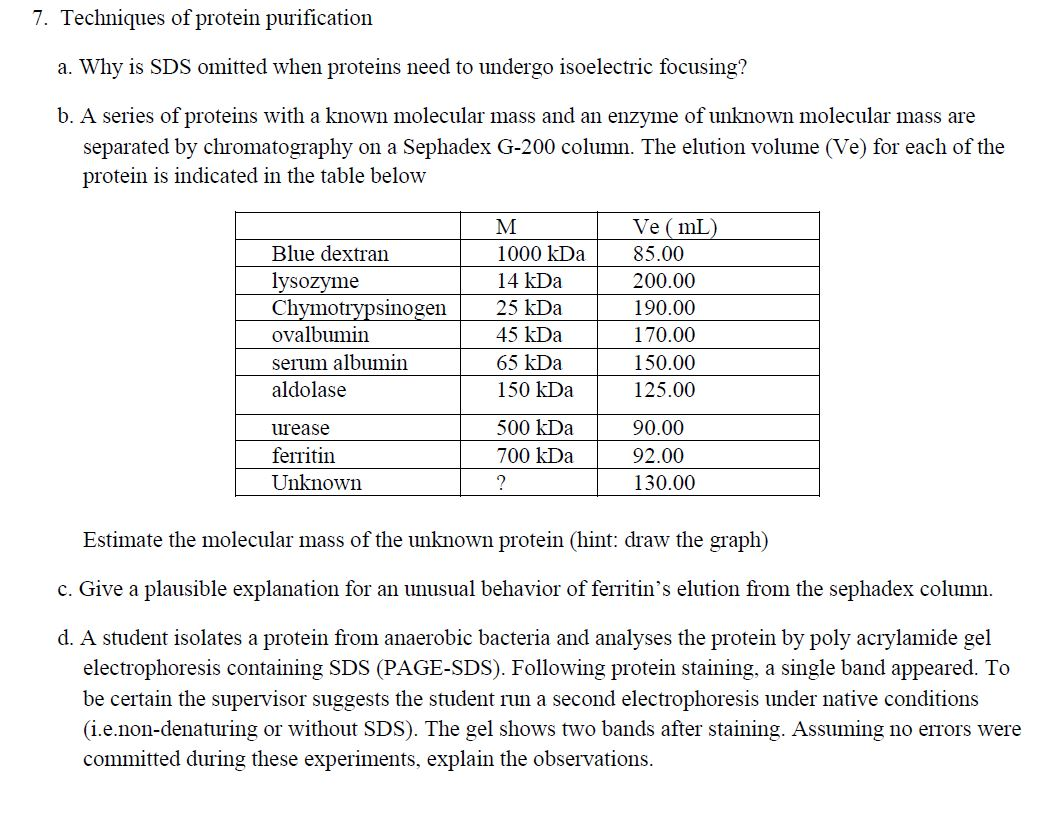
7. Techniques of protein purification a. Why is SDS omitted when proteins need to undergo isoelectric focusing? b. A series of proteins with a known molecular mass and an enzyme of unknown molecular mass are separated by chromatography on a Sephadex G-200 column. The elution volume (Ve) for each of the protein is indicated in the table below Blue dextran lysozyme Chymotrypsinogen ovalbumin serum albumin aldolase urease ferritin Unknown M 1000 kDa 14 kDa 25 kDa 45 kDa 65 kDa 150 kDa 500 kDa 700 kDa ? Ve (mL) 85.00 200.00 190.00 170.00 150.00 125.00 90.00 92.00 130.00 Estimate the molecular mass of the unknown protein (hint: draw the graph) c. Give a plausible explanation for an unusual behavior of ferritin's elution from the sephadex column. d. A student isolates a protein from anaerobic bacteria and analyses the protein by poly acrylamide gel electrophoresis containing SDS (PAGE-SDS). Following protein staining, a single band appeared. To be certain the supervisor suggests the student run a second electrophoresis under native conditions (i.e.non-denaturing or without SDS). The gel shows two bands after staining. Assuming no errors were committed during these experiments, explain the observations. 7. Techniques of protein purification a. Why is SDS omitted when proteins need to undergo isoelectric focusing? b. A series of proteins with a known molecular mass and an enzyme of unknown molecular mass are separated by chromatography on a Sephadex G-200 column. The elution volume (Ve) for each of the protein is indicated in the table below Blue dextran lysozyme Chymotrypsinogen ovalbumin serum albumin aldolase urease ferritin Unknown M 1000 kDa 14 kDa 25 kDa 45 kDa 65 kDa 150 kDa 500 kDa 700 kDa ? Ve (mL) 85.00 200.00 190.00 170.00 150.00 125.00 90.00 92.00 130.00 Estimate the molecular mass of the unknown protein (hint: draw the graph) c. Give a plausible explanation for an unusual behavior of ferritin's elution from the sephadex column. d. A student isolates a protein from anaerobic bacteria and analyses the protein by poly acrylamide gel electrophoresis containing SDS (PAGE-SDS). Following protein staining, a single band appeared. To be certain the supervisor suggests the student run a second electrophoresis under native conditions (i.e.non-denaturing or without SDS). The gel shows two bands after staining. Assuming no errors were committed during these experiments, explain the observations.
Expert Answer:
Answer rating: 100% (QA)
Answer 7a Isoelectric point is that pH when the proteins net charge is zero ie in electrically neutral state The purpose of the isoelectric focusing i... View the full answer

Related Book For 

Biochemistry Concepts and Connections
ISBN: 978-0321839923
1st edition
Authors: Dean R. Appling, Spencer J. Anthony-Cahill, Christopher K. Mathews
Posted Date:
Students also viewed these biology questions
-
A protein gives a single band on SDS gel electrophoresis, as shown in lanes 1 and 2 below. There is little if any effect from adding β -mercaptoethanol (BME) to the sample; if anything,...
-
When proteins are separated using native gel electrophoresis, size, shape, and charge control their rate of migration on the gel. Why does DNA separate based on size, and why do we not worry much...
-
Under ideal conditions a certain bacteria population is known to double every three hours. Suppose that there are initially 100 bacteria. (a) What is the size of the population after 15 hours? (b)...
-
Find a and b if, y= ax+b has mean 6 and variance is unity, where x is a random variable with mean 8 and variance 16.
-
Why do we study the reversed Carnot cycle even though it is not a realistic model for refrigeration cycles?
-
Express the area of the region bounded by x = 4 - 4y 2 , y = 1, and x = 2 in at least two different ways, one of which is an iterated integral. Do not find the area of the region.
-
Repeat all parts of Problem 15.D12 but with a water rate of \(q=0.000015 \mathrm{~m}^{2} / \mathrm{s}\). Problem 15.D12 Water at \(20^{\circ} \mathrm{C}\) is flowing down a \(3.0 \mathrm{~m}\) long...
-
During its first year of operations, Sugarsmooth, Inc. produced 55,000 jars of hand cream based on a formula containing 10 percent glycolic acid. Unit sales were 53,500 jars. Fixed overhead totaled...
-
Match the description with the highest level of measurement the data has - The names of various flowers are gathered and recorded - The dates of a solar eclipse are recorded - The top grossing movies...
-
The Porsche Club of America sponsors driver education events that provide high-performance driving instruction on actual race tracks. Because safety is a primary consideration at such events, many...
-
Topics and key terms have ranged from classical to contemporary and have included behavior, style, traits, job-centered,employee-centered, concern for people, concern for production, motivation,...
-
What is natural selection, and how does it work? Give an example of natural selection in action.
-
Explain the methodology and findings of the following empirical studies, examining the B-S OPM: a. Black-Scholes (1972) b. Galai (1977) c. MacBeth and Merville (1979)
-
Give some examples of unique human linguistic abilities.
-
Explain how a company planning to issue four-year, fixed-rate bonds in two years could use a forward swap to lock in the fix rate it will pay on the bonds. Explain how the hedge works at the...
-
Suppose you are studying chimp communication. What limitations will you have in teaching the chimps a human language?
-
Calculate the 95% confidence interval for mean total intake of sodium over the 14 days. Round final answers to nearest whole number. [Hint: Section 20.3] Include any formulas including numerical...
-
The water in tank A is at 270 F with quality of 10% and mass 1 lbm. It is connected to a piston/cylinder holding constant pressure of 40 psia initially with 1 lbm water at 700 F. The valve is opened,...
-
Carbon dioxide is dissolved in blood (pH 7.4) to form a mixture of carbonic acid and bicarbonate. Neglecting free CO2, what fraction will be present as carbonic acid? Would you expect a signicant...
-
Radioactive uracil can be used to label all of the pyrimidine residues in DNA. Using either names or structures, present pathways for the conversion of uracil to dTTP and to dCTP for each reaction,...
-
Te activity of an enzyme requires a glutamic acid to display its - COOH functional group in the protonated state. Suppose the pKa of the - COOH group is 4.07 a. Will the enzyme be more active at pH...
-
Use Aspen Plus Analysis to develop and print out ternary diagrams for the system water-chloroform-acetone using NRTL as the LLE correlation. Compare results to experimental data in Table 13-A2 (mole...
-
Do Lab 12 in this chapter's appendix, but for three-stage systems. Operation is at \(293 \mathrm{~K}, 1.0 \mathrm{~atm}, \mathrm{~F}=10 \mathrm{kmol} / \mathrm{h}\) and is \(10 \mathrm{~mol} \%\)...
-
Input the DECHEMA coefficients in Aspen Plus from Table 13-A1 in the appendix. Then in Properties, have Aspen print out the triangular diagram for the triethylamine, carbon-tetrachloride, acetic-acid...

Study smarter with the SolutionInn App


