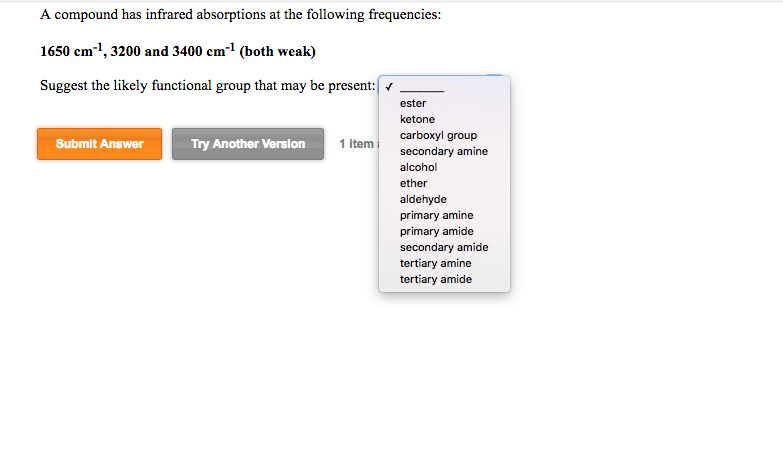
A compound has infrared absorptions at the following frequencies: 1650 cm, 3200 and 3400 cm (both...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A compound has infrared absorptions at the following frequencies: 1650 cm¹¹, 3200 and 3400 cm¹ (both weak) Suggest the likely functional group that may be present: ✔ ester ketone carboxyl group secondary amine alcohol ether Submit Answer Try Another Version 1 Item aldehyde primary amine primary amide secondary amide tertiary amine tertiary amide A compound has infrared absorptions at the following frequencies: 1650 cm¹¹, 3200 and 3400 cm¹ (both weak) Suggest the likely functional group that may be present: ✔ ester ketone carboxyl group secondary amine alcohol ether Submit Answer Try Another Version 1 Item aldehyde primary amine primary amide secondary amide tertiary amine tertiary amide
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Compound A, C 4 H 8 O 3 , has infrared absorptions at 1710 and 2500 to 3100 cm ?1 and has the 1 H NMR spectrum shown. Propose a structure for A. 11.18 TMS 10 O ppm 8. 6. 3. Chemical shift (6)...
-
List at least five risks that may be present in a company, and that may be associated with material misstatements in the company's financial statements. Does the existence of one or more of these...
-
help A compound has infrared absorptions at the following frequencies: 3300 and 3385 cm (both weak) and no band between 1600 and 1850 cm Suggest the likely functional group that may be preser Submit...
-
Suppose that two stocks whose rates of return are given by the two-factor arbitrage pricing model r= a +31 +42 r2 = 02 +241 +2, where a, and a2 are constants. Furthermore, there is a risk-free asset...
-
Discuss the strategic implications of marketing in Mexico.
-
Here the stone is sliding down a friction-free incline. (a) Identify the forces that act on it, and draw appropriate force vectors, (b) By the parallelogram rule, construct the resultant force on the...
-
You have started a job at an annual salary of \($32,000\). Your take-home pay is about 2/3 of your gross salary.
-
Wash Clean Laboratories produces biodegradable liquid detergents that leave no soap film. The production process has been automated, so the product can now be produced in one operation instead of in...
-
Risk and Return Homework Hotels A and B have the following probability distributions of expected future returns: Economy Probability Hotel A Hotel B Recession 30% (20%) 20% Stable 40% 15% 25% Boom...
-
The information listed below refers to the employees of Lemonica Company for the year ended December 31, 2016. The wages are separated into the quarters in which they were paid to the individual...
-
You think that the stock of Fleetwood Corp is likely to rise within the next six months from its current price ($29.00 bid and $30.00 ask), and you want to maximize the amount of profit from your...
-
At which value(s) for x does the graph of y = x-3x - 10x + 7 have horizontal tangent lines?
-
Question 3 of 10 Wildhorse Enterprises provides the following information relative to its defined benefit pension plan. Balances or Values at December 31, 2025 Projected benefit obligation $2,727,900...
-
Delph Company uses job-order costing with a plantwide predetermined overhead rate based on machine-hours. At the beginning of the year, the company estimated that 50,000 machine-hours would be...
-
If a Hershey's Chocolate allows its lower level managers to aid in the decision making process because these managers are more aware of what is going on and need to develop their decision making...
-
Primare Corporation provided the following data for last month's manufacturing operations. Purchases of raw materials Indirect materials used in production Direct labor Manufacturing overhead applied...
-
Hey, single card is drawn at random from a deck of playing cards. Find the probability the card is an ace or numbered card. Round to the hundredths
-
The electric field due to a line charge is given by where l is a constant. Show that E is solenoidal. Show that it is also conservative. E =
-
Show the structure, including stereochemistry, of the product from the following Diels?Alder reaction: COCH3 C
-
Treatment of a cyclic ketone with diazomethane is a method for accomplishing a ring-expansion reaction. For example, treatment of Cyclohexanone with diazomethane yields Cycloheptanone. Propose...
-
We?ll see that there are two isomeric substances both named 1, 2-dimethylcyclohexane. Explain. -C3 1,2-Dimethylcyclohexane CH
-
To become a member of an Internet DVD library, Maritza has to put down a deposit of \($100,\) using her credit card. When and if Maritza chooses to discontinue her membership, the library will refund...
-
If your personal financial statements indicate that you have a deficit, what might you do to change your financial situation?
-
How do you calculate your net worth when preparing a balance sheet?

Study smarter with the SolutionInn App



