A compressed air tank in a mechanic shop holds a volume of 1 m of air...
Fantastic news! We've Found the answer you've been seeking!
Question:
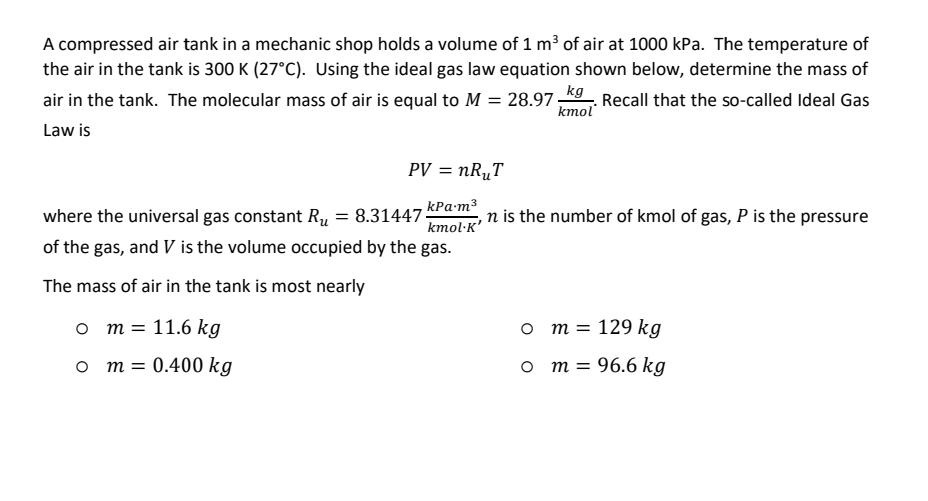
Transcribed Image Text:
A compressed air tank in a mechanic shop holds a volume of 1 m³ of air at 1000 kPa. The temperature of the air in the tank is 300 K (27°C). Using the ideal gas law equation shown below, determine the mass of air in the tank. The molecular mass of air is equal to M = 28.97 kg. Recall that the so-called Ideal Gas Law is kmol PV = nR₁T kPa.m³ kmol·K' where the universal gas constant R₁ = 8.31447- of the gas, and V is the volume occupied by the gas. The mass of air in the tank is most nearly O m = 11.6 kg O m = 0.400 kg n is the number of kmol of gas, P is the pressure O m = 129 kg ○ m = 96.6 kg A compressed air tank in a mechanic shop holds a volume of 1 m³ of air at 1000 kPa. The temperature of the air in the tank is 300 K (27°C). Using the ideal gas law equation shown below, determine the mass of air in the tank. The molecular mass of air is equal to M = 28.97 kg. Recall that the so-called Ideal Gas Law is kmol PV = nR₁T kPa.m³ kmol·K' where the universal gas constant R₁ = 8.31447- of the gas, and V is the volume occupied by the gas. The mass of air in the tank is most nearly O m = 11.6 kg O m = 0.400 kg n is the number of kmol of gas, P is the pressure O m = 129 kg ○ m = 96.6 kg
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below The problem statemen... View the full answer

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
Let A, B be sets. Define: (a) the Cartesian product (A B) (b) the set of relations R between A and B (c) the identity relation A on the set A [3 marks] Suppose S, T are relations between A and B, and...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
A local university is considering changes to its class structure in an effort to increase professor productivity. The old schedule had each professor teaching 5 classes per week, with each class...
-
Dirtworks Machinery Company is an authorized distributor of heavy machinery and equipment produced by Caterpillar, Inc. On February 15, 2009, Dirtworks purchased 10 backhoe loaders from Caterpillar...
-
a. Find the local extrema of each function on the given interval, and say where they occur. b. Graph the function and its derivative together. Comment on the behavior of in relation to the signs and...
-
After returning from a skiing vacation in Vermont, Leslie Adel came down with Legionnaires Disease. He claimed it was from the water drunk at the ski resort and provided by Greensprings of Vermont...
-
The East Eanes School District engaged in or was affected by the following events and transactions during its scal year ending June 30, 2015. 1. Teachers and other personnel earned $350,000 in...
-
Consider again the market studied in Question 2, The demand curve is given by P = 100 Q and the marginal cost curve is given by MC = 10. This time we are going to assume that firms must invest funds...
-
The accompanying table shows a portion of data consisting of the January, April, July, and October average temperatures of 50 selected U.S. cities. a. Perform agglomerative hierarchical clustering to...
-
Should U.S. companies be penalized for importing their own products from other countries. Although Ford is complying with the letter of the law, are Fords actions proper? Give 2 major reasons...
-
Eddie Green owns and operates a small business Paradise Garden that sells gardening products through its online store. All sales revenue received from online sales is recorded as unearned revenue at...
-
What are the main ideas of this article? Chap-2 Customer Service (SCM) Exhibit: Poor Service Can Result in Lost Customers AT&T is one of the largest Internet and cell phone providers in the United...
-
1. (2 pts) An "OR" gate can be used to "OR" two bits together. Explain how it can be used to enable/disable another digital signal. 2. (2 pts) Explain how an "OR" gate can be used to set bits in a...
-
Consider the DTMC below: -5 So === cifen ciles 121 9 1 $4 1+1=1414 1|1 12 $5 0/0 Let A = {s3} and B = {s2}. 1. Compute the probability measure of the union of the following cylinder sets: Cyl(sos1),...
-
Capacitance of plate capacitor C = 3 . 5 nF and the gap between the plates d = 1 . 3 mm . The capacitor is attached to a voltage source whose voltage is V 0 = 2 9 V . a ) How big is the electric...
-
Suppose that the constant q is such that no horizontal line can be tangent to the curve y = 2r+3qx?+6x+131 at any point. Then (a) q > -2 (b) q < 2 (c) -2 < q < 2 (d) q < -2 or q > 2 (e) none of these
-
During the month, services performed for customers on account amounted to $7,500 and collections from customers in payment of their accounts totaled $6,000. At the end of the month, the Accounts...
-
Pharmacokinetics concerns the ingestion, distribution, reaction, and elimination reaction of drugs in the body. Consider the application of pharmacokinetics to one of the major problems we have in...
-
The gas-phase reaction 12N2+32H2NH3 is to be carried out isothermally first in a flow reactor. The molar feed is 50% H 2 and 50% N 2 , at a pressure of 16.4 atm and at a temperature of 227C. a....
-
Repeat parts (a)(c) of Problem P4-11B for the reaction 3A C Pure A enters at 400 K, 10 atm and the equilibrium constant is K C = 2.5 (dm 3 /mol) 2 . Compare the equilibrium conversions in Problems...
-
Sharing on various digital media impulsively can lead to embarrassment and worse. Have you or has someone you know ever regretted posting a comment, photo, or other digital media online? What...
-
Explain how communication skills fuel career success, and understand why writing skills are vital in a digital, mobile, and social-mediadriven workplace.
-
What employability skills are Canadian employers looking for in an employee?

Study smarter with the SolutionInn App


